Advertisements
Advertisements
Question
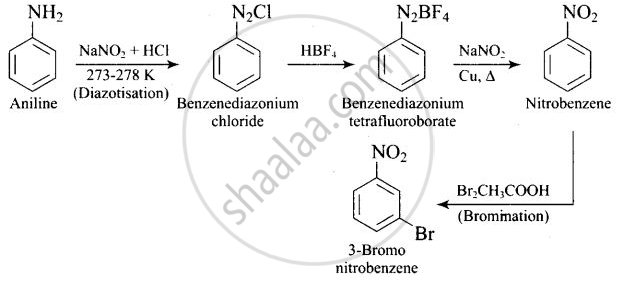
How will you carry out the following conversion?

Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Give the structures of A, B and C in the following reactions :

Write the reaction of aromatic primary amine with nitrous acid.
Write the reaction of aliphatic primary amine with nitrous acid.
Write structures of compounds A and B in each of the following reactions:
Answer in one sentence.
Which amide does produce ethanamine by Hofmann bromamide degradation reaction?
The following amines is the product of Gabriel phthalimide synthesis.
Identify the product 'A' in the following reaction.
\[\ce{Aniline ->[(CH3CO)2O][Pyridine] A}\]
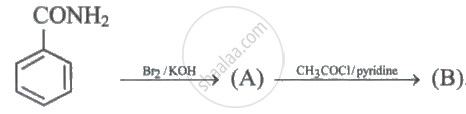
Identify the major product (B).
Which of the following reactions is appropriate for converting benzamide to aniline?
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is ______.
Hoffmann Bromamide Degradation reaction is shown by ______.
Acetamide and ethyl amide can be distinguished by reacting with.
Write a short note on Ammonolysis.
Write short note on the following:
Ammonolysis
Write a short note on Ammonolysis.
