Advertisements
Advertisements
प्रश्न
Give a plausible explanation for the following:
Why are amines less acidic than alcohols of comparable molecular masses?
Advertisements
उत्तर
Loss of a proton from an amine gives an amide ion, while loss of a proton from alcohol gives an alkoxide ion.
\[\ce{R-NH2 -> R-NH– + H+}\]
\[\ce{R-O-H -> R-O^- + H+}\]
Since O is more electronegative than N, it will attract positive species more strongly in comparison to N. Thus, RO− is more stable than RNH−. Thus, alcohols are more acidic than amines. Conversely, amines are less acidic than alcohols.
APPEARS IN
संबंधित प्रश्न
Identify the compounds 'A' and 'B' in the following equation:

Give the structures of A, B and C in the following reactions :

Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Mention 'two' uses of propan-2-one.
Give the structures of A, B and C in the following reactions :
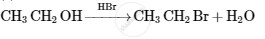
Mendius reaction is used to convert _____________
Why cannot aniline be prepared by Gabriel phthalimide synthesis?
Which of the following does NOT give carbylamine test?
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds:
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

How will you carry out the following conversion?

Which of the following reactions will not give a primary amine?
Write short note on the following.
Ammonolysis.
