Advertisements
Advertisements
Question
Amongst the following, the strongest base in aqueous medium is ______.
Options
\[\ce{CH3NH2}\]
\[\ce{NCCH2NH2}\]
\[\ce{(CH3)2NH}\]
\[\ce{C6H5NHCH3}\]
Advertisements
Solution
Amongst the following, the strongest base in aqueous medium is \[\ce{(CH3)2NH}\].
Explanation:
2° amine is more basic than 1° amine, i.e., \[\ce{(CH3)2NH}\] is more basic than \[\ce{CH3NH2}\]. Due to \[\ce{^-I}\] effect of \[\ce{CN}\] group, \[\ce{NC-CH2NH2}\] is less basic than \[\ce{CH3NH2}\]. Further \[\ce{C6H5NHCH3}\] is less basic than both \[\ce{CH3NH2}\] and \[\ce{(CH3)2NH}\] due to delocalization of lone pair of electrons present on the nitrogen atom into benzene ring. Hence, the decreasing order of amines is:
\[\ce{(CH3)2NH > CH3NH2 > C6H5NHCH3 > NC – CH2NH2}\]
APPEARS IN
RELATED QUESTIONS
Accomplish the following conversion:
Nitrobenzene to benzoic acid
Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Accomplish the following conversion:
Benzamide to toluene
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :

Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Give the structures of A, B and C in the following reactions :

Choose the most correct option.
Which of the following compounds will dissolve in aqueous NaOH after undergoing reaction with Hinsberg reagent?
Write reactions for the preparation of ethanamine using Gabriel phthalimide synthesis.
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
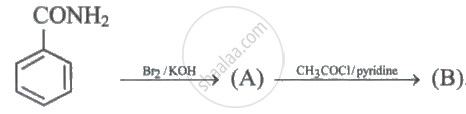
Identify the major product (B).
Which of the following reactions is appropriate for converting benzamide to aniline?
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
What is the best reagent to convert nitrile to primary amine?
How will you carry out the following conversion?

How will you carry out the following conversions?

Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write a short note on the following:
Ammonolysis.
