Advertisements
Advertisements
Question
Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Advertisements
Solution
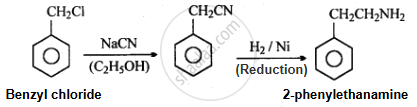
APPEARS IN
RELATED QUESTIONS
How are propan-1-amine and propan-2-amine prepared from oxime?
Identify the compounds 'A' and 'B' in the following equation:

How do you convert the following: C6H5CONH2 to C6H5NH2
Give the structures of A, B and C in the following reactions :

Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2 + HCl][273 K] B ->[H2O/H+][\Delta] C}\]
Write the reaction of aliphatic primary amine with nitrous acid.
Answer in one sentence.
Predict the product of the following reaction.
\[\ce{Nitrobenzene ->[Sn/conc.HCl]?}\]
Answer the following
Explain Gabriel phthalimide synthesis.
Write the order of reactivity of alkyl halides with ammonia.
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
Quaternary ammonium salt is formed:
The source of nitrogen in Gabriel synthesis of amines is ______.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is ______.
Write following conversions:
nitrobenzene `->` acetanilide
Describe Gabriel's phthalimide synthesis. (Give reaction)
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
