Advertisements
Advertisements
Question
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2 + HCl][273 K] B ->[H2O/H+][\Delta] C}\]
Advertisements
Solution
\[\ce{\underset{Nitrobenzene}{C6H5NO2} ->[Fe/HCl] \underset{(A)}{\underset{Aniline}{C6H5NH2}} ->[NaNO2 + HCl][273 K] \underset{(B)}{\underset{chloride}{\underset{Benzenediazonium}{C6H5\overset{+}{N}≡NCl-}}} ->[H2O/H+][\Delta] \underset{(C)}{\underset{Phenol}{C6H5OH}}}\]
APPEARS IN
RELATED QUESTIONS
How is ethyl amine prepared from methyl iodide?
Accomplish the following conversion:
Benzamide to toluene
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Acetamide on reduction using Na/C2H5OH gives ____________.
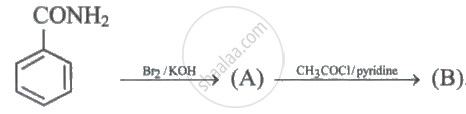
Identify the major product (B).
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
Identify the product obtained when benzamide is treated with bromine and aqueous sodium hydroxide?
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds:
Among the following amines, the strongest Brönsted base is:
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Write short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis.
Write a short note on the following:
Ammonolysis
