Advertisements
Advertisements
Question
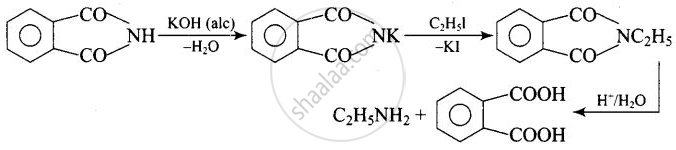
The source of nitrogen in Gabriel synthesis of amines is ______.
Options
Sodium azide, \[\ce{NaN3}\]
Sodium nitrite, \[\ce{NaNO2}\]
Potassium cyanide, \[\ce{KCN}\]
Potassium phthalimide, \[\ce{C6H4(CO)2N– K+}\]
Advertisements
Solution
The source of nitrogen in Gabriel synthesis of amines is Potassium phthalimide, \[\ce{C6H4(CO)2N– K+}\].
Explanation:
Potassium phthalimide is the source of nitrogen in Gabriel’s synthesis.
APPEARS IN
RELATED QUESTIONS
Write a short note on the following:
Hoffmann’s bromamide reaction
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :

Answer the following
Explain the ammonolysis of alkyl halides.
Write the order of reactivity of alkyl halides with ammonia.
Write reactions to bring about the following conversions.
Acetamide to Ethylamine
Write reactions to bring about the following conversions.
Acetamide to methylamine
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
In aqueous phase the order of basic strength of alkylamine is ______.
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds:
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Alkyl halides are insoluble in water.
Reason (R): Alkyl halides have halogen attached to sp3 hybrid carbon.
Select the most appropriate answer from the options given below:
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
Ethylamine can be prepared by the action of bromine and caustic potash on which compound?
In the given reaction what is the X?
\[\begin{array}{cc}
\ce{O}\phantom{.......................}\\
||\phantom{.......................}\\
\phantom{}\ce{R - C - OH <-[H3O] Χ ->[H] RCH2NH2}
\end{array}\]
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
Write short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis
Write short notes on the following:
Ammonolysis
