Advertisements
Advertisements
प्रश्न
The source of nitrogen in Gabriel synthesis of amines is ______.
विकल्प
Sodium azide, \[\ce{NaN3}\]
Sodium nitrite, \[\ce{NaNO2}\]
Potassium cyanide, \[\ce{KCN}\]
Potassium phthalimide, \[\ce{C6H4(CO)2N– K+}\]
Advertisements
उत्तर
The source of nitrogen in Gabriel synthesis of amines is Potassium phthalimide, \[\ce{C6H4(CO)2N– K+}\].
Explanation:
Potassium phthalimide is the source of nitrogen in Gabriel’s synthesis.
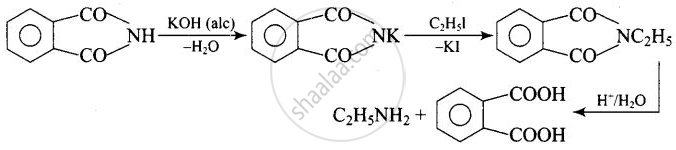
APPEARS IN
संबंधित प्रश्न
Give the structures of A, B and C in the following reactions :

Accomplish the following conversion:
Nitrobenzene to benzoic acid
Give the structures of A, B and C in the following reactions :
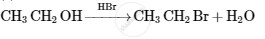
Account for the following:
Gabriel phthalimide synthesis is not preferred for preparing aromatic primary amines.
The following amines is the product of Gabriel phthalimide synthesis.
Write reactions to bring about the following conversions.
Acetamide to Ethylamine
Write reactions to bring about the following conversions.
Acetamide to methylamine
Acetamide on reduction using Na/C2H5OH gives ____________.
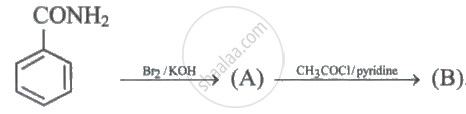
Identify the major product (B).
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
Which of the following compounds is obtained when quaternary ammonium hydroxide is strongly heated?
Benzylamine may be alkylated as shown in the following equation:
\[\ce{C6H5CH2NH2 + R - X -> C6H5CH2NHR}\]
Which of the following alkylhalides is best suited for this reaction through SN1 mechanism?
Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
The Gabriels' phthalimide synthesis is used in the synthesis of
Which of the following reactions will not give a primary amine?
Ethylamine can be prepared by the action of bromine and caustic potash on which compound?
Which of the following amines can be prepared by Gabriel phthalimide reaction?
Identify the compo ds A and B in the following reactions:
\[\ce{A ->[Nitrating mixture] B ->[(i) Sn/cone. HCI][(ii) NaOH] Aniline}\]
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write a short note on Ammonolysis.
