Advertisements
Advertisements
प्रश्न
The source of nitrogen in Gabriel synthesis of amines is ______.
पर्याय
Sodium azide, \[\ce{NaN3}\]
Sodium nitrite, \[\ce{NaNO2}\]
Potassium cyanide, \[\ce{KCN}\]
Potassium phthalimide, \[\ce{C6H4(CO)2N– K+}\]
Advertisements
उत्तर
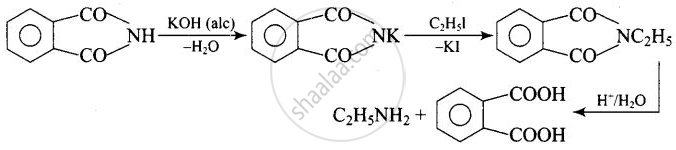
The source of nitrogen in Gabriel synthesis of amines is Potassium phthalimide, \[\ce{C6H4(CO)2N– K+}\].
Explanation:
Potassium phthalimide is the source of nitrogen in Gabriel’s synthesis.
APPEARS IN
संबंधित प्रश्न
Identify the compounds 'A' and 'B' in the following equation:

Give the structures of A, B and C in the following reactions :
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
____________ can be prepared exclusively by Gabriel phthalimide synthesis.
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
Which of the following reagents is used in Hofmann's elimination reaction of amines?
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is ______.
Which of the following compounds is the weakest Brönsted base?
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
How will you bring out the following conversion?

How will you carry out the following conversions?

The compound X is which of the following?
\[\ce{CH3CN ->[Na + C2H5OH] x}\]
Acetamide and ethyl amide can be distinguished by reacting with.
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
