Advertisements
Advertisements
प्रश्न
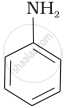
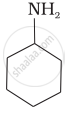
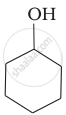
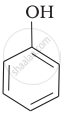
Which of the following compounds is the weakest Brönsted base?
पर्याय
Advertisements
उत्तर
Explanation:
Amines (i, ii) have stronger tendency to accept a proton and hence are stronger Bronsted bases than phenol (iii) and alcohol (iv). Since phenol is more acidic than alcohol, therefore, phenol (iii) has the least tendency to accept a proton and hence it is the weakest Bronsted base.
APPEARS IN
संबंधित प्रश्न
How is ethyl amine prepared from methyl iodide?
Give the structures of A, B and C in the following reaction:
\[\ce{CH3COOH ->[NH3][\Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\]
Account for the following:
Gabriel phthalimide synthesis is not preferred for preparing aromatic primary amines.
Answer the following
Identify A and B in the following reactions.
\[\ce{C6H5CH2Br->[alco.][KCN]A ->[Na/ethanol]B.}\]
Answer the following
Explain Gabriel phthalimide synthesis.
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Explain the following reaction with a suitable example.
Hofmann elimination reaction
Identify the product obtained, when benzamide is treated with bromine and aqueous sodium hydroxide.
____________ can be prepared exclusively by Gabriel phthalimide synthesis.
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Amongst the following, the strongest base in aqueous medium is ______.
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Write following conversions:
nitrobenzene `->` acetanilide
Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
A primary amine is formed by an amide on treatment with bromine and alkali. The primary amine has
Write the name of the product formed by the action of LiAlH4/ether on acetamide.
Write short note on the following:
Ammonolysis