Advertisements
Advertisements
प्रश्न
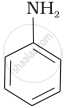
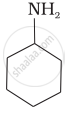
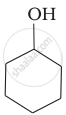
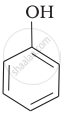
Which of the following compounds is the weakest Brönsted base?
विकल्प
Advertisements
उत्तर
Explanation:
Amines (i, ii) have stronger tendency to accept a proton and hence are stronger Bronsted bases than phenol (iii) and alcohol (iv). Since phenol is more acidic than alcohol, therefore, phenol (iii) has the least tendency to accept a proton and hence it is the weakest Bronsted base.
APPEARS IN
संबंधित प्रश्न
How do you convert the following: Ethanenitrile to ethanamine
Write a short note on the following:
Hoffmann’s bromamide reaction
Write the reaction of aliphatic primary amine with nitrous acid.
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Answer the following
Identify A and B in the following reactions.
\[\ce{C6H5CH2Br->[alco.][KCN]A ->[Na/ethanol]B.}\]
Write reactions for the preparation of ethanamine using Gabriel phthalimide synthesis.
Identify the product 'A' in the following reaction.
\[\ce{Aniline ->[(CH3CO)2O][Pyridine] A}\]
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
Which of the following reactions is appropriate for converting benzamide to aniline?
In aqueous phase the order of basic strength of alkylamine is ______.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Alkyl halides are insoluble in water.
Reason (R): Alkyl halides have halogen attached to sp3 hybrid carbon.
Select the most appropriate answer from the options given below:
Benzylamine may be alkylated as shown in the following equation:
\[\ce{C6H5CH2NH2 + R - X -> C6H5CH2NHR}\]
Which of the following alkylhalides is best suited for this reaction through SN1 mechanism?
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Identify A and B in the following reaction.

Write following conversions:
nitrobenzene `->` acetanilide
Assertion: Aromatic 1° amines can be prepared by Gabriel Phthalimide Synthesis.
Reason: Aryl halides undergo nucleophilic substitution with anion formed by phthalimide.
Identify the compo ds A and B in the following reactions:
\[\ce{A ->[Nitrating mixture] B ->[(i) Sn/cone. HCI][(ii) NaOH] Aniline}\]
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write a short note on the following:
Ammonolysis