Advertisements
Advertisements
प्रश्न
Among the following amines, the strongest Brönsted base is:
विकल्प
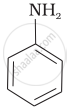
\[\ce{NH3}\]
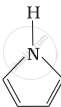
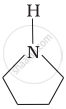
Advertisements
उत्तर

Explanation:
Pyrrolidine is the strongest of two bases because the lone pair of nitrogen does not involve sin resonance and the presence of two alkyl basic compounds increases the basic strength among the given four compounds.
APPEARS IN
संबंधित प्रश्न
How are propan-1-amine and propan-2-amine prepared from oxime?
Give a plausible explanation for the following:
Why are amines less acidic than alcohols of comparable molecular masses?
Account for the following:
Gabriel phthalimide synthesis is not preferred for preparing aromatic primary amines.
Write structures of compounds A and B in each of the following reactions:
Write reactions to bring about the following conversions.
Acetamide to methylamine
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
Which of the following compounds is obtained when quaternary ammonium hydroxide is strongly heated?
Which of the following reactions does NOT yield an amine?
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Reduction of nitro alkanes yields which compound?
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write short note on the following:
Ammonolysis
