Advertisements
Advertisements
प्रश्न
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
Advertisements
उत्तर
(i) Isobutyl amine
(ii) 2-Phenylethylamine
Explanation:
Gabriel synthesis is a method for producing primary amines. When phthalimide is treated with ethanolic potassium hydroxide, it forms a potassium salt of phthalimide, which when heated with an alkyl halide and then alkaline hydrolyzed yields the corresponding primary amine.
APPEARS IN
संबंधित प्रश्न
Give the structures of A, B and C in the following reactions :

Write the reaction of aromatic primary amine with nitrous acid.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Give the structures of A, B and C in the following reactions :

Mendius reaction is used to convert _____________
Write reactions to bring about the following conversions.
Acetamide to methylamine
Why cannot aniline be prepared by Gabriel phthalimide synthesis?
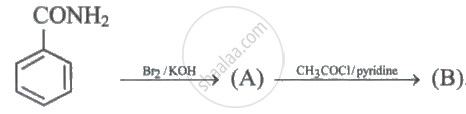
Identify the major product (B).
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
In aqueous phase the order of basic strength of alkylamine is ______.
The source of nitrogen in Gabriel synthesis of amines is ______.
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Reduction of nitrobenzene by which of the following reagent gives aniline?
(i) \[\ce{Sn/HCl}\]
(ii) \[\ce{Fe/HCl}\]
(iii) \[\ce{H2 - Pd}\]
(iv) \[\ce{Sn/NH4OH}\]
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc.\[\ce{H2SO4 }\] + conc. \[\ce{HNO3}\].
(ii) Acetic anyhdride/pyridine followed by conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iii) Dil. HCl followed by reaction with conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iv) Reaction with conc.\[\ce{HNO3}\] + conc.\[\ce{H2 SO4}\].
The Gabriels' phthalimide synthesis is used in the synthesis of
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
Write the name of the product formed by the action of LiAlH4/ether on acetamide.
Write a short note on the following:
Ammonolysis.
