Advertisements
Advertisements
Question
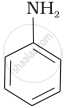
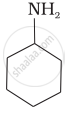
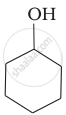
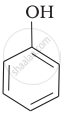
Which of the following compounds is the weakest Brönsted base?
Options
Advertisements
Solution
Explanation:
Amines (i, ii) have stronger tendency to accept a proton and hence are stronger Bronsted bases than phenol (iii) and alcohol (iv). Since phenol is more acidic than alcohol, therefore, phenol (iii) has the least tendency to accept a proton and hence it is the weakest Bronsted base.
APPEARS IN
RELATED QUESTIONS
Write the order of reactivity of alkyl halides with ammonia.
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
What product is formed when \[\ce{R - C ≡ N}\] is hydrolysed?
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Which of the following does NOT give carbylamine test?
Amongst the following, the strongest base in aqueous medium is ______.
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Write following conversions:
nitrobenzene `->` acetanilide
How will you carry out the following conversions?

When primary amines are treated with HCl, the product obtained is which of the following?
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
Write a short note on the following:
Ammonolysis