Advertisements
Advertisements
Question
Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc.\[\ce{H2SO4 }\] + conc. \[\ce{HNO3}\].
(ii) Acetic anyhdride/pyridine followed by conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iii) Dil. HCl followed by reaction with conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iv) Reaction with conc.\[\ce{HNO3}\] + conc.\[\ce{H2 SO4}\].
Advertisements
Solution
(i) Acetyl chloride/pyridine followed by reaction with conc.\[\ce{H2SO4 }\] + conc. \[\ce{HNO3}\].
(ii) Acetic anyhdride/pyridine followed by conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
Explanation:

APPEARS IN
RELATED QUESTIONS
How do you convert the following: C6H5CONH2 to C6H5NH2
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH + Br2] C}\]
Give the structures of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[HNO2][273 K] B ->[C6H5OH] C}\]
Give a plausible explanation for the following:
Why are amines less acidic than alcohols of comparable molecular masses?
Give the structures of A, B and C in the following reactions :
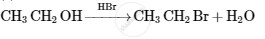
Answer the following
Explain Gabriel phthalimide synthesis.
Mendius reaction is used to convert _____________
Identify the product obtained when benzamide is treated with bromine and aqueous sodium hydroxide?
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Write following conversions:
nitrobenzene `->` acetanilide
Write following conversions:
acetanilide `->` p-nitroaniline
In the given reaction what is the X?
\[\begin{array}{cc}
\ce{O}\phantom{.......................}\\
||\phantom{.......................}\\
\phantom{}\ce{R - C - OH <-[H3O] Χ ->[H] RCH2NH2}
\end{array}\]
Give reasons for the following:
Ammonolysis of alkyl halides is not a good method to prepare pure primary amines.
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Write a short note on the following:
Ammonolysis
Write a short note on Ammonolysis.
Write a short note on Ammonolysis.
