Advertisements
Advertisements
Question
Why does acetylation of –NH2 group of aniline reduce its activating effect?
Advertisements
Solution
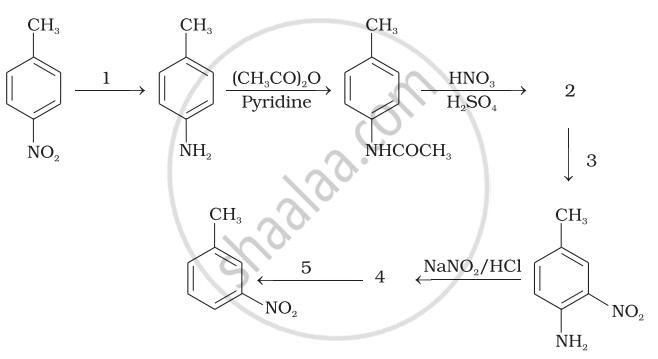
Direct nitration of aniline is not possible on account of oxidation of –NH2 group. However, nitration can be carried after protecting the –NH2 group by acetylation to give acetanilide which is then nitrated and finally hydrolysed to give o- and p-nitroanilines.
The acetyl group being electron-withdrawing attracts the lone pair of electrons of the N-atom towards carbonyl group.

As a result, the activating effect –NH2 group is reduced i.e., the lone pair of electrons on nitrogen is less available for donation to benzene ring by resonance. Therefore, activating effect of –NHCOCH3 group is less than that of –NH2 group.
APPEARS IN
RELATED QUESTIONS
Write equations of the following reactions:
Acetylation of aniline
Arenium ion involved in the bromination of aniline is:
(i)

(ii)

(iii)

(iv)

Why is benzene diazonium chloride not stored and is used immediately after its preparation?
Predict the product of reaction of aniline with bromine in non-polar solvent such as \[\ce{CS2}\].
Why is aniline soluble in aqueous HCl?
How will you carry out the following conversions?
toluene `->` p-toluidine
How will you carry out the following conversions?
p-toluidine diazonium chloride `→` p-toluic acid
Match the compounds given in Column I with the items given in Column II.
| Column I | Column II | ||
| (i) | Benzene sulphonyl chloride | (a) | Zwitter ion |
| (ii) | Sulphanilic acid | (b) | Hinsberg reagent |
| (iii) | Alkyl diazonium salts | (c) | Dyes |
| (iv) | Aryl diazonium salts | (d) | Conversion to alcohols |
Predict the reagent or the product in the following reaction sequence.
In order to distinguish between C2H5NHz and C6H5NHz, which of the following reagents is useful?
Aniline when treated with cone. HNO3 gives
Which of the following is the most stable diazonium salt?
Benzene diazonium chloride is a ______.
Consider the following compounds:
(i) p-methyl aniline
(ii) N, N-dimethylaniline
(iii) N-ethyl aniline
(iv) N-ethyl-N-methyl aniline
The compounds which do not form diazonium salt with ice-colds NaNO2 and HCl are:
Aniline dissolved in dilute HCl is reacted with sodium nitrate at 0 °C. This solution was added dropwise to a solution containing equimolar mixture of aniline and phenol in dil. HCl. The structure of the major product is:
Identify A and B for the following reaction:

What are polymers?
How will the following be converted? (Give chemical equation)
Aniline to benzene diazonium chloride.
