Advertisements
Advertisements
Question
Write equations of the following reactions:
Acetylation of aniline
Advertisements
Solution

APPEARS IN
RELATED QUESTIONS
Illustrate the following reaction giving suitable example in each case: Diazotisation
Why is aniline soluble in aqueous HCl?
Match the compounds given in Column I with the items given in Column II.
| Column I | Column II | ||
| (i) | Benzene sulphonyl chloride | (a) | Zwitter ion |
| (ii) | Sulphanilic acid | (b) | Hinsberg reagent |
| (iii) | Alkyl diazonium salts | (c) | Dyes |
| (iv) | Aryl diazonium salts | (d) | Conversion to alcohols |
Predict the reagent or the product in the following reaction sequence.
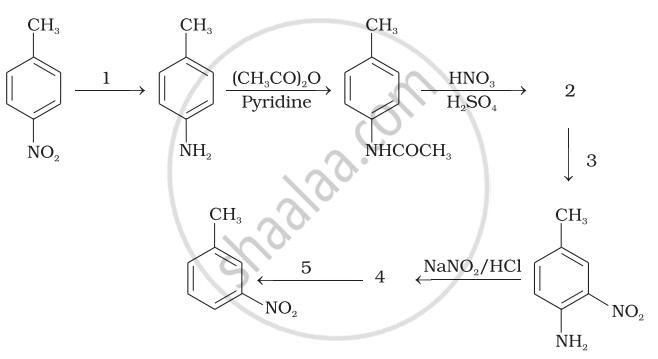
Aniline when treated with cone. HNO3 gives
Which of the following is true characteristic feature of aniline?
Which of the following is the most stable diazonium salt?
Benzene diazonium chloride is a ______.
Consider the following compounds:
(i) p-methyl aniline
(ii) N, N-dimethylaniline
(iii) N-ethyl aniline
(iv) N-ethyl-N-methyl aniline
The compounds which do not form diazonium salt with ice-colds NaNO2 and HCl are:
Aniline dissolved in dilute HCl is reacted with sodium nitrate at 0 °C. This solution was added dropwise to a solution containing equimolar mixture of aniline and phenol in dil. HCl. The structure of the major product is:
