Advertisements
Advertisements
Question
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Options
Reaction of nitrite with \[\ce{LiAlH4}\].
Reaction of amide with \[\ce{LiAlH4}\] followed by treatment with water
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis.
Treatment of amide with bromine in aqueous solution of sodium hydroxide.
Advertisements
Solution
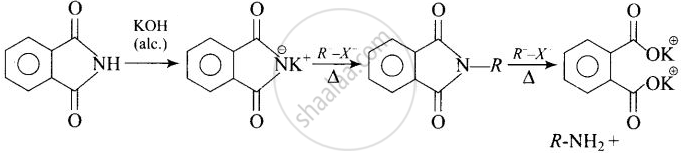
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis.
Explanation:
Aliphatic and arylalkyl primary amines can be prepared by the reduction of the corresponding nitriles with \[\ce{LiAlH4}\].
\[\ce{\underset{Alkynitrite}{R - C ≡ N} \underset{Arynitrite}{Ar - C ≡ N} -> LiAlH4 RCH2\underset{1° amine}{NH2} or ArCh2NH2}\]
Heating alkyl halide with primary, secondary and tertiary amine can be prepared by reduction of \[\ce{LiAlH4}\] followed by treatment with water.
\[\ce{\underset{1° amide}{R - CONH2} ->[(i) LiAlH4][(ii) H2O] R - CH2 - NH2}\]
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis produces primary amine. This process is known as Gabriel phthalimide reaction. The number of carbon atoms in the chain of amines of product is same as reactant.
APPEARS IN
RELATED QUESTIONS
Mention 'two' uses of propan-2-one.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Write structures of compounds A and B in each of the following reactions:
Why cannot aniline be prepared by Gabriel phthalimide synthesis?
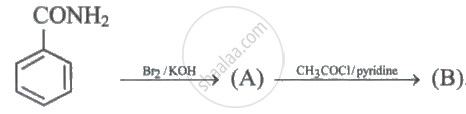
Identify the major product (B).
Which of the following reactions is appropriate for converting benzamide to aniline?
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
The source of nitrogen in Gabriel synthesis of amines is ______.
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is ______.
Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc.\[\ce{H2SO4 }\] + conc. \[\ce{HNO3}\].
(ii) Acetic anyhdride/pyridine followed by conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iii) Dil. HCl followed by reaction with conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iv) Reaction with conc.\[\ce{HNO3}\] + conc.\[\ce{H2 SO4}\].
Suggest a route by which the following conversion can be accomplished.

The Gabriels' phthalimide synthesis is used in the synthesis of
Reduction of nitro alkanes yields which compound?
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write a short note on the following:
Ammonolysis
