Advertisements
Advertisements
प्रश्न
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
पर्याय
Reaction of nitrite with \[\ce{LiAlH4}\].
Reaction of amide with \[\ce{LiAlH4}\] followed by treatment with water
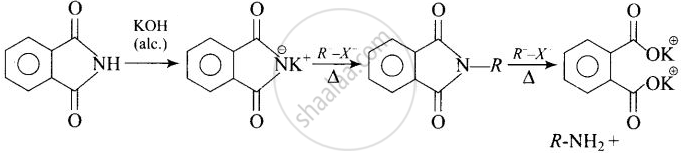
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis.
Treatment of amide with bromine in aqueous solution of sodium hydroxide.
Advertisements
उत्तर
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis.
Explanation:
Aliphatic and arylalkyl primary amines can be prepared by the reduction of the corresponding nitriles with \[\ce{LiAlH4}\].
\[\ce{\underset{Alkynitrite}{R - C ≡ N} \underset{Arynitrite}{Ar - C ≡ N} -> LiAlH4 RCH2\underset{1° amine}{NH2} or ArCh2NH2}\]
Heating alkyl halide with primary, secondary and tertiary amine can be prepared by reduction of \[\ce{LiAlH4}\] followed by treatment with water.
\[\ce{\underset{1° amide}{R - CONH2} ->[(i) LiAlH4][(ii) H2O] R - CH2 - NH2}\]
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis produces primary amine. This process is known as Gabriel phthalimide reaction. The number of carbon atoms in the chain of amines of product is same as reactant.
APPEARS IN
संबंधित प्रश्न
Write a short note on the following:
Hoffmann’s bromamide reaction
Accomplish the following conversion:
Nitrobenzene to benzoic acid
Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Write the reaction of aliphatic primary amine with nitrous acid.
Give the structures of A, B and C in the following reactions :

Answer in one sentence.
Predict the product of the following reaction.
\[\ce{Nitrobenzene ->[Sn/conc.HCl]?}\]
Answer the following
Write a reaction to convert acetic acid into methylamine.
Name the process of breaking C-X bond by ammonia in preparation of amines.
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Quaternary ammonium salt is formed:
Among the following amines, the strongest Brönsted base is:
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write short notes on the following:
Ammonolysis
