Advertisements
Advertisements
प्रश्न
Write reactions for the preparation of ethanamine using Gabriel phthalimide synthesis.
Advertisements
उत्तर
Gabriel phthalimide synthesis for ethanamine:
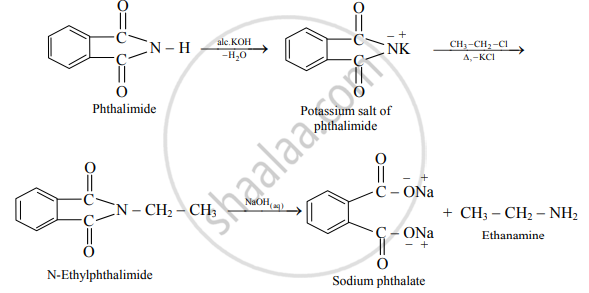
APPEARS IN
संबंधित प्रश्न
Write the chemical equation involved in the following reaction:
Hoffmann-bromamide degradation reaction
Give the structures of A, B and C in the following reactions :

Write a short note on the following:
Hoffmann’s bromamide reaction
Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Accomplish the following conversion:
Benzamide to toluene
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH + Br2] C}\]
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2Br ->[KCN] A ->[LiAlH4] B ->[HNO2][0^\circ C] C}\]
Give the structures of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[HNO2][273 K] B ->[C6H5OH] C}\]
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :

Give the structures of A, B and C in the following reactions :

Give the structures of A, B and C in the following reactions :

Answer the following
Identify A and B in the following reactions.
\[\ce{C6H5CH2Br->[alco.][KCN]A ->[Na/ethanol]B.}\]
Answer the following
Write a reaction to convert acetic acid into methylamine.
Answer the following
Explain Gabriel phthalimide synthesis.
Answer the following
Explain the ammonolysis of alkyl halides.
Name the process of breaking C-X bond by ammonia in preparation of amines.
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
Identify the product obtained, when benzamide is treated with bromine and aqueous sodium hydroxide.
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
What product is formed when \[\ce{R - C ≡ N}\] is hydrolysed?
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Identify the product obtained when benzamide is treated with bromine and aqueous sodium hydroxide?
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Which of the following compounds is obtained when quaternary ammonium hydroxide is strongly heated?
For producing amines, the reaction of nitro compounds with iron scrap is preferred because:
Amongst the following, the strongest base in aqueous medium is ______.
Benzylamine may be alkylated as shown in the following equation:
\[\ce{C6H5CH2NH2 + R - X -> C6H5CH2NHR}\]
Which of the following alkylhalides is best suited for this reaction through SN1 mechanism?
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is ______.
Which of the following compounds is the weakest Brönsted base?
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
What is the best reagent to convert nitrile to primary amine?
Identify A and B in the following reaction.

Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
The Gabriels' phthalimide synthesis is used in the synthesis of
Acetamide and ethyl amide can be distinguished by reacting with.
When primary amines are treated with HCl, the product obtained is which of the following?
Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Amides can be converted into amines by the reaction named ______.
Write short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis
Write short note on the following:
Ammonolysis
Write short note on the following.
Ammonolysis.
Assertion: Amimonolysis of alkyl halides involves the reaction between alkyl halides and alcoholic ammonia.
Reason: Ammonolysis of alkyl halides produces secondary amines only.
Write a short note on the following:
Ammonolysis
