Advertisements
Advertisements
प्रश्न
Answer the following
Explain the ammonolysis of alkyl halides.
Advertisements
उत्तर
i. When alkyl halide is heated with an alcoholic solution of excess ammonia, it undergoes a nucleophilic substitution reaction in which the halogen atom is replaced by an amino (–NH2) group to form primary amine.
ii. This process of breaking of the C – X bond by ammonia is known as ammonolysis. The reaction is also known as the alkylation of ammonia. The reaction is carried out in a sealed tube at 373 K.
iii. The primary amine obtained in the1st step is stronger nucleophile than ammonia. Hence, it further reacts with an alkyl halide to form secondary and tertiary amines and finally quaternary ammonium salt if NH3 is not used in large excess.
\[\ce{\underset{\text{Alkyl halide}}{R - X} + \underset{\text{(excess)}}{NH3_{(alc.)}}->[\Delta] \underset{1^0}{R} - NH2 + HX}\]
iv. The order of reactivity of alkyl halides with ammonia is R–I > R–Br > R–Cl.
APPEARS IN
संबंधित प्रश्न
Give the structures of A, B and C in the following reactions :

Accomplish the following conversion:
Nitrobenzene to benzoic acid
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2Br ->[KCN] A ->[LiAlH4] B ->[HNO2][0^\circ C] C}\]
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2 + HCl][273 K] B ->[H2O/H+][\Delta] C}\]
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Write the reaction of aliphatic primary amine with nitrous acid.
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Mention 'two' uses of propan-2-one.
Give the structures of A, B and C in the following reactions :
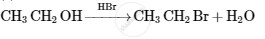
Give the structures of A, B and C in the following reactions :

Give the structures of A, B and C in the following reactions :

Account for the following:
Gabriel phthalimide synthesis is not preferred for preparing aromatic primary amines.
Write structures of compounds A and B in each of the following reactions:
Answer in one sentence.
Which amide does produce ethanamine by Hofmann bromamide degradation reaction?
Answer the following
Identify A and B in the following reactions.
\[\ce{C6H5CH2Br->[alco.][KCN]A ->[Na/ethanol]B.}\]
Answer the following
Write a reaction to convert acetic acid into methylamine.
Name the process of breaking C-X bond by ammonia in preparation of amines.
Acetamide on reduction using Na/C2H5OH gives ____________.
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
Identify the product obtained, when benzamide is treated with bromine and aqueous sodium hydroxide.
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
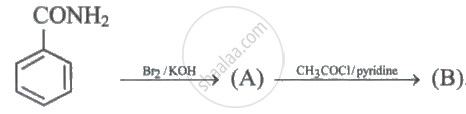
Identify the major product (B).
____________ can be prepared exclusively by Gabriel phthalimide synthesis.
What product is formed when \[\ce{R - C ≡ N}\] is hydrolysed?
Identify 'A' and 'B' in the following conversions.
\[\ce{CH3 - I ->[Alc. KCN][\Delta] A ->[Na/C2H5OH] B}\]
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds:
For producing amines, the reaction of nitro compounds with iron scrap is preferred because:
Amongst the following, the strongest base in aqueous medium is ______.
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
Hoffmann Bromamide Degradation reaction is shown by ______.
Which of the following compounds is the weakest Brönsted base?
Among the following amines, the strongest Brönsted base is:
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
Give reasons for the following:
Ammonolysis of alkyl halides is not a good method to prepare pure primary amines.
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
Which of the following amines can be prepared by Gabriel phthalimide reaction?
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
What is the IUPAC name of \[\ce{(CH3)2 - N - CH3}\]?
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Amides can be converted into amines by the reaction named ______.
Write a short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis.
