Advertisements
Advertisements
प्रश्न
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
पर्याय
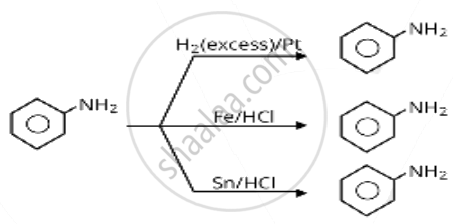
\[\ce{H2 (excess)/Pt}\]
\[\ce{LiAlH4}\] in ether
\[\ce{Fe}\] and \[\ce{HCl}\]
\[\ce{Sn}\] and \[\ce{HCl}\]
Advertisements
उत्तर
\[\ce{LiAlH4}\] in ether
Explanation:
Lithium aluminium hydride in either is a strong reducing agent that donated its \[\ce{H-}\] hydride ion to any \[\ce{C = O}\] containing a functional group. In addition to \[\ce{LiAlH}\], to aryl nitro compounds, no reaction will be observed, the desired products of amines will not be produced, rather it will form diazobenzene products.
\[\ce{2C4H5NO ->[LiAlH4/ether] C6H5N = N - C6H5}\]
APPEARS IN
संबंधित प्रश्न
How are propan-1-amine and propan-2-amine prepared from oxime?
Illustrate the following reaction giving suitable example in each case:Gabriel phthalimide synthesis
Answer the following
Write a reaction to convert acetic acid into methylamine.
Answer the following
Explain Gabriel phthalimide synthesis.
Identify the product 'A' in the following reaction.
\[\ce{Aniline ->[(CH3CO)2O][Pyridine] A}\]
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Benzylamine may be alkylated as shown in the following equation:
\[\ce{C6H5CH2NH2 + R - X -> C6H5CH2NHR}\]
Which of the following alkylhalides is best suited for this reaction through SN1 mechanism?
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is ______.
Which of the following compounds is the weakest Brönsted base?
Among the following amines, the strongest Brönsted base is:
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
Which of the following CANNOT be prepared by ammonolysis of alkyl halide?
Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Write the name of the product formed by the action of LiAlH4/ether on acetamide.
