Advertisements
Advertisements
प्रश्न
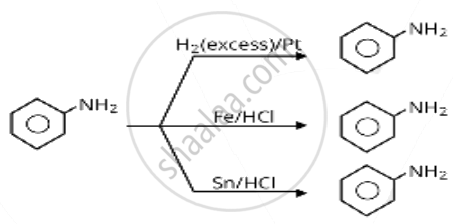
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
विकल्प
\[\ce{H2 (excess)/Pt}\]
\[\ce{LiAlH4}\] in ether
\[\ce{Fe}\] and \[\ce{HCl}\]
\[\ce{Sn}\] and \[\ce{HCl}\]
Advertisements
उत्तर
\[\ce{LiAlH4}\] in ether
Explanation:
Lithium aluminium hydride in either is a strong reducing agent that donated its \[\ce{H-}\] hydride ion to any \[\ce{C = O}\] containing a functional group. In addition to \[\ce{LiAlH}\], to aryl nitro compounds, no reaction will be observed, the desired products of amines will not be produced, rather it will form diazobenzene products.
\[\ce{2C4H5NO ->[LiAlH4/ether] C6H5N = N - C6H5}\]
APPEARS IN
संबंधित प्रश्न
How is ethyl amine prepared from methyl iodide?
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2Br ->[KCN] A ->[LiAlH4] B ->[HNO2][0^\circ C] C}\]
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :

Give the structures of A, B and C in the following reactions :

Choose the most correct option.
Which of the following compounds will dissolve in aqueous NaOH after undergoing reaction with Hinsberg reagent?
Mendius reaction is used to convert _____________
Write reactions to bring about the following conversions.
Acetamide to methylamine
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Identify 'A' and 'B' in the following conversions.
\[\ce{CH3 - I ->[Alc. KCN][\Delta] A ->[Na/C2H5OH] B}\]
Which of the following amines cannot be prepared by Gabriel phthalimide synthesis?
Amongst the following, the strongest base in aqueous medium is ______.
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Write a short note on the following:
Ammonolysis
Write short note on the following.
Ammonolysis.
Write a short note on Ammonolysis.
