Advertisements
Advertisements
प्रश्न
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Advertisements
उत्तर

Mechanism:
Step-1:
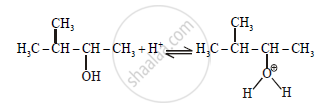
Step-2:
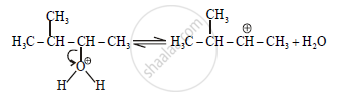
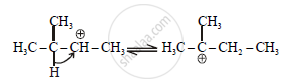
Step-3:
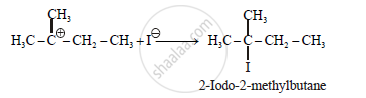
APPEARS IN
संबंधित प्रश्न
How is ethyl amine prepared from methyl iodide?
Write the chemical equation involved in the following reaction:
Hoffmann-bromamide degradation reaction
Accomplish the following conversion:
Nitrobenzene to benzoic acid
Accomplish the following conversion:
Benzamide to toluene
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH + Br2] C}\]
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Give the structures of A, B and C in the following reaction:
\[\ce{CH3COOH ->[NH3][\Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\]
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :

Answer in one sentence.
Which amide does produce ethanamine by Hofmann bromamide degradation reaction?
Answer in one sentence.
Predict the product of the following reaction.
\[\ce{Nitrobenzene ->[Sn/conc.HCl]?}\]
Answer the following
Write a reaction to convert acetic acid into methylamine.
The following amines is the product of Gabriel phthalimide synthesis.
Mendius reaction is used to convert _____________
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Explain the following reaction with a suitable example.
Hofmann elimination reaction
Identify the product 'A' in the following reaction.
\[\ce{Aniline ->[(CH3CO)2O][Pyridine] A}\]
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
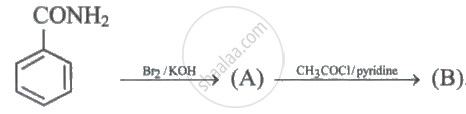
Identify the major product (B).
____________ can be prepared exclusively by Gabriel phthalimide synthesis.
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Which of the following reactions is appropriate for converting benzamide to aniline?
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Identify 'A' and 'B' in the following conversions.
\[\ce{CH3 - I ->[Alc. KCN][\Delta] A ->[Na/C2H5OH] B}\]
Which of the following amines cannot be prepared by Gabriel phthalimide synthesis?
Which of the following does NOT give carbylamine test?
Benzylamine may be alkylated as shown in the following equation:
\[\ce{C6H5CH2NH2 + R - X -> C6H5CH2NHR}\]
Which of the following alkylhalides is best suited for this reaction through SN1 mechanism?
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Write following conversions:
nitrobenzene `->` acetanilide
How will you carry out the following conversions?

Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
Reduction of nitro alkanes yields which compound?
Which of the following compound is expected to be most basic?
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
Which of the following compound gives pink colour on reaction with phthalic anhydride in cone. H2SO4 followed by treatment with NaOH?
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
Which of the following amines can be prepared by Gabriel phthalimide reaction?
What is the IUPAC name of \[\ce{(CH3)2 - N - CH3}\]?
Write the name of the product formed by the action of LiAlH4/ether on acetamide.
Amides can be converted into amines by the reaction named ______.
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
Write short note on the following:
Ammonolysis
Write short note on the following.
Ammonolysis.
Write short notes on the following:
Ammonolysis
