Advertisements
Advertisements
प्रश्न
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
Advertisements
उत्तर
Ethyl cyanide (or Propanenitrile) \[\ce{C2H5 - CN}\] after treatment with sodium and alcohol (ethanol) produces n-propyl amine.
\[\ce{\underset{(Ethyl cyanide)}{CH3 - CH2 - CN} ->[Na/C2H5OH] \underset{(n-Propyl amine)}{CH3 - CH2 - CH2 - NH2}}\]
The above reduction is known as the Mendius reduction.
APPEARS IN
संबंधित प्रश्न
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Write a short note on the following:
Hoffmann’s bromamide reaction
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Give the structures of A, B and C in the following reaction:
\[\ce{CH3COOH ->[NH3][\Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\]
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Write the reaction of aromatic primary amine with nitrous acid.
Give the structures of A, B and C in the following reactions :
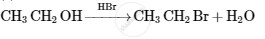
Write structures of compounds A and B in each of the following reactions:
Answer the following
Identify A and B in the following reactions.
\[\ce{C6H5CH2Br->[alco.][KCN]A ->[Na/ethanol]B.}\]
Answer the following
Explain Gabriel phthalimide synthesis.
Mendius reaction is used to convert _____________
Write reactions to bring about the following conversions.
Acetamide to Ethylamine
Write reactions to bring about the following conversions.
Acetamide to methylamine
Explain the following reaction with a suitable example.
Hofmann elimination reaction
Acetamide on reduction using Na/C2H5OH gives ____________.
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Identify the product obtained when benzamide is treated with bromine and aqueous sodium hydroxide?
Identify 'A' and 'B' in the following conversions.
\[\ce{CH3 - I ->[Alc. KCN][\Delta] A ->[Na/C2H5OH] B}\]
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Which of the following does NOT give carbylamine test?
Quaternary ammonium salt is formed:
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Alkyl halides are insoluble in water.
Reason (R): Alkyl halides have halogen attached to sp3 hybrid carbon.
Select the most appropriate answer from the options given below:
Hoffmann Bromamide Degradation reaction is shown by ______.
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

Suggest a route by which the following conversion can be accomplished.

Identify A and B in the following reaction.

Write following conversions:
acetanilide `->` p-nitroaniline
How will you bring out the following conversion?

How will you carry out the following conversion?

Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
The Gabriels' phthalimide synthesis is used in the synthesis of
Reduction of nitro alkanes yields which compound?
Which of the following amines can be prepared by Gabriel phthalimide reaction?
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
What is the IUPAC name of \[\ce{(CH3)2 - N - CH3}\]?
Write the name of the product formed by the action of LiAlH4/ether on acetamide.
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write short note on the following:
Ammonolysis
Write short note on the following:
Ammonolysis
Write a short note on Ammonolysis.
Write short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis
