Advertisements
Advertisements
प्रश्न
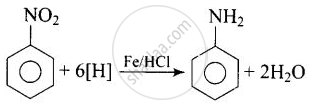
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
पर्याय
aromatic oxime
aromatic hydrocarbon
aromatic primary amine
aromatic amide
Advertisements
उत्तर
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives aromatic primary amine.
Explanation:
APPEARS IN
संबंधित प्रश्न
How is ethyl amine prepared from methyl iodide?
Write the chemical equation involved in the following reaction:
Hoffmann-bromamide degradation reaction
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Give the structures of A, B and C in the following reactions :

Write the reaction of aliphatic primary amine with nitrous acid.
Answer in one sentence.
Which amide does produce ethanamine by Hofmann bromamide degradation reaction?
Answer in one sentence.
Predict the product of the following reaction.
\[\ce{Nitrobenzene ->[Sn/conc.HCl]?}\]
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds:
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Suggest a route by which the following conversion can be accomplished.

How will you carry out the following conversion?

How will you carry out the following conversions?

Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
The compound X is which of the following?
\[\ce{CH3CN ->[Na + C2H5OH] x}\]
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
Which of the following CANNOT be prepared by ammonolysis of alkyl halide?
Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
