Advertisements
Advertisements
प्रश्न
The most reactive amine towards dilute hydrochloric acid is:
पर्याय
CH3 – NH2
\[\begin{array}{cc}
\ce{H3C}\phantom{.............}\\\
\backslash\phantom{.......}\\
\ce{NH}\\
/\phantom{.......}\\
\ce{H3C}\phantom{...............}
\end{array}\]\[\begin{array}{cc}
\ce{H3C}\phantom{..........................}\\
\backslash\phantom{..................}\\
\ce{NH - CH3}\phantom{}\\
/\phantom{...................}\\
\ce{H3C}\phantom{...........................}\\
\end{array}\]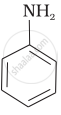
Advertisements
उत्तर
\[\begin{array}{cc}
\ce{H3C}\phantom{.............}\\\
\backslash\phantom{.......}\\
\ce{NH}\\
/\phantom{.......}\\
\ce{H3C}\phantom{...............}
\end{array}\]
Explanation:
The greater the strength of a base, the greater will be its reactivity towards dilute HCl. Hence, (CH3)2NH has the highest basic strength as it has the highest reactivity.
