Advertisements
Advertisements
प्रश्न
Write the reaction of aromatic primary amine with nitrous acid.
Advertisements
उत्तर
Aromatic amine react with nitrous acid (prepared in situ from NaNO2 and a mineral acid such as HCl) at 273 − 278 K to form stable aromatic diazonium salts, i.e., NaCl and H2O.

APPEARS IN
संबंधित प्रश्न
Write a short note on Hoffmann bromamide degradation.
Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Account for the following:
Gabriel phthalimide synthesis is not preferred for preparing aromatic primary amines.
Write reactions to prepare ethanamine from Acetonitrile.
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Write reactions for the preparation of ethanamine using Gabriel phthalimide synthesis.
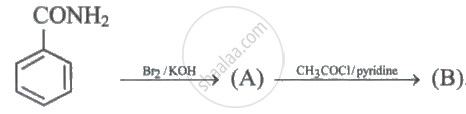
Identify the major product (B).
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Which of the following compounds is obtained when quaternary ammonium hydroxide is strongly heated?
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds:
For producing amines, the reaction of nitro compounds with iron scrap is preferred because:
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Identify A and B in the following reaction.

How will you carry out the following conversion?

Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
Identify the compo ds A and B in the following reactions:
\[\ce{A ->[Nitrating mixture] B ->[(i) Sn/cone. HCI][(ii) NaOH] Aniline}\]
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
