Advertisements
Advertisements
प्रश्न
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Advertisements
उत्तर
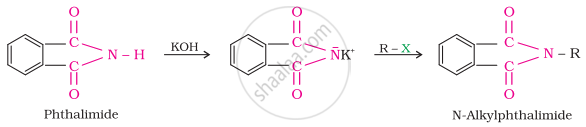
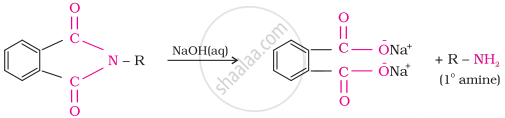
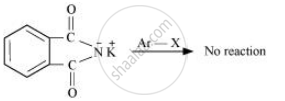
Gabriel synthesis is used for the preparation of primary amines. Phthalimide, on treatment with ethanolic potassium hydroxide, forms potassium salt of phthalimide, which on heating with alkyl halide followed by alkaline hydrolysis produces the corresponding primary amine. Aromatic primary amines cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide.
APPEARS IN
संबंधित प्रश्न
Give the structures of A, B and C in the following reactions :

Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2Br ->[KCN] A ->[LiAlH4] B ->[HNO2][0^\circ C] C}\]
Give a plausible explanation for the following:
Why are amines less acidic than alcohols of comparable molecular masses?
Account for the following:
Gabriel phthalimide synthesis is not preferred for preparing aromatic primary amines.
Write reactions to prepare ethanamine from Acetonitrile.
The following amines is the product of Gabriel phthalimide synthesis.
Mendius reaction is used to convert _____________
Write the order of reactivity of alkyl halides with ammonia.
Identify the product 'A' in the following reaction.
\[\ce{Aniline ->[(CH3CO)2O][Pyridine] A}\]
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Quaternary ammonium salt is formed:
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
Among the following amines, the strongest Brönsted base is:
How will you bring out the following conversion?

Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
Write short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis
