Advertisements
Advertisements
प्रश्न
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Advertisements
उत्तर
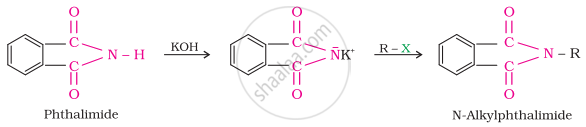
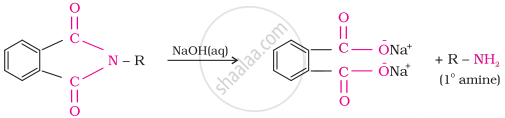
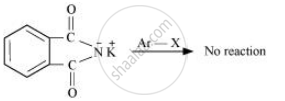
Gabriel synthesis is used for the preparation of primary amines. Phthalimide, on treatment with ethanolic potassium hydroxide, forms potassium salt of phthalimide, which on heating with alkyl halide followed by alkaline hydrolysis produces the corresponding primary amine. Aromatic primary amines cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide.
APPEARS IN
संबंधित प्रश्न
How are propan-1-amine and propan-2-amine prepared from oxime?
Give the structures of A, B and C in the following reactions :

Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Write structures of compounds A and B in each of the following reactions:
Which of the following reactions is appropriate for converting benzamide to aniline?
Which of the following amines cannot be prepared by Gabriel phthalimide synthesis?
Which of the following compounds is obtained when quaternary ammonium hydroxide is strongly heated?
Which of the following reactions does NOT yield an amine?
The source of nitrogen in Gabriel synthesis of amines is ______.
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

How will you bring out the following conversion?

Which of the following reactions will not give a primary amine?
The compound X is which of the following?
\[\ce{CH3CN ->[Na + C2H5OH] x}\]
Which of the following compound is expected to be most basic?
Which of the following compound gives pink colour on reaction with phthalic anhydride in cone. H2SO4 followed by treatment with NaOH?
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
Write a short note on Ammonolysis.
