Advertisements
Advertisements
प्रश्न
Aniline does not give Friedel-Crafts reaction. Give a reason.
Advertisements
उत्तर
Aniline does not undergo Friedel-Craft's reaction because the reaction takes place in the presence of AlCl3, but AlCl3 is a Lewis acid, whereas aniline is a strong basic. Thus, aniline interacts with AlCl3 to produce a salt.
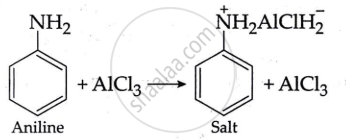
The positive charge on the N-atom prevents electrophilic substitution in the benzene ring. As a result, aniline does not undergo the Friedel-Craft reaction.
APPEARS IN
संबंधित प्रश्न
How is chlorobenzene prepared from aniline?
What is the action of acetic anhydride on ethylamine?
What is the action of the following reagents on aniline?
Bromine water
Account for the following:
Although the amino group is o, p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
Write short notes on acetylation.
What is the action of acetic anhydride on diethylamine?
How will you convert the following?
Aniline into N−phenylethanamide
What is the role of \[\ce{HNO3}\] in the nitrating mixture used for nitration of benzene?
A solution contains 1 g mol. each of p-toluene diazonium chloride and p-nitrophenyl diazonium chloride. To this 1 g mol. of alkaline solution of phenol is added. Predict the major product. Explain your answer.
Give reasons for the following observation:
Aniline does not react with methyl chloride in the presence of anhydrous AlCl3 catalyst.
