Advertisements
Advertisements
Question
Write the reaction involved in the Stephen reduction
Advertisements
Solution
Stephen reduction: In this reaction, nitriles are reduced to corresponding imine with stannous chloride in the presence of hydrochloric acid, which on hydrolysis give corresponding aldehyde.
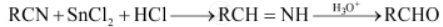
shaalaa.com
Is there an error in this question or solution?
