Advertisements
Advertisements
Questions
How will you bring about the following conversion?
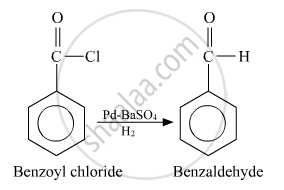
Benzoyl chloride to benzaldehyde.
How will you convert the following: (Give a balanced equation)
Benzoyl chloride to benzaldehyde.
Chemical Equations/Structures
Advertisements
Solution 1
shaalaa.com
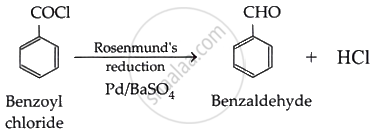
Solution 2
Benzoyl chloride can be converted to benzaldehyde by carrying out Rosenmund’s reaction:
shaalaa.com
Is there an error in this question or solution?
2012-2013 (March) Delhi Set 1
