Advertisements
Chapters
1: Solid State
2: Solutions
3: Electrochemistry
4: Chemical Kinetics
5: Surface Chemistry
Inorganic Chemistry
6: General Principles and Processes of Isolation of Elements
7: p-Block Elements
8: d-and ƒ-Block Elements
9: Coordination Compounds
Organic Chemistry
▶ 10: Haloalkanes and Haloarenes
Chapter 11: Alcohols, Phenols and Ethers
Chapter 12: Aldehydes, Ketones and Carboxylic Acids
Chapter 13: Organic Compounds containing Nitrogen
Chapter 14: Biomolecules
15: Polymers
Chapter 16: Chemistry in Everyday Life
![Nootan solutions for Chemistry [English] Class 12 ISC chapter 10 - Haloalkanes and Haloarenes Nootan solutions for Chemistry [English] Class 12 ISC chapter 10 - Haloalkanes and Haloarenes - Shaalaa.com](/images/chemistry-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Advertisements
Solutions for Chapter 10: Haloalkanes and Haloarenes
Below listed, you can find solutions for Chapter 10 of CISCE Nootan for Chemistry [English] Class 12 ISC.
Nootan solutions for Chemistry [English] Class 12 ISC 10 Haloalkanes and Haloarenes REVIEW EXERCISES [Pages 567 - 604]
What are haloalkanes?
How are haloalkanes classified?
What do you understand by a 3° alkyl halide?
Give an example of a 3° alkyl halide.
What is the main structural difference between 1° and 2° alkyl halides?
Write the structure of the following:
Vinyl chloride
Write the structure of the following:
Allyl chloride
Write the structure of a chloroalkene.
Write the structure of a chloroalkyne.
Write the structural formula of the following compound.
n-propyl chloride
Write the IUPAC name of the following compound.
n-propyl chloride
Write the structural formula of the following compound.
iso-butyl chloride
Write the IUPAC name of the following compound.
iso-butyl chloride
Write the structural formula of the following compound.
sec-butyl chloride
Write the IUPAC name of the following compound.
sec-butyl chloride
Write the structural formula of the following compound.
iso-amyl chloride
Write the IUPAC name of the following compound.
iso-amyl chloride
Write the structural formula of the following compound.
neo-pentyl chloride
Write the IUPAC name of the following compound.
neo-pentyl chloride
Give the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{\phantom{.}F\phantom{..........}Cl}\\
|\phantom{...........}|\\
\ce{CH3 - C - CH2 - C - CH3}\\
|\phantom{...........}|\\
\ce{\phantom{....}C2H5\phantom{......}C2H5}\\
\end{array}\]
Give the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{\phantom{.........}CH3}\\
\phantom{.......}|\\
\ce{CH3 - CH - C - CH3}\\
\phantom{..}|\phantom{......}|\phantom{..}\\
\ce{\phantom{...}Cl\phantom{....}CH3}\\
\end{array}\]
Give the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{\phantom{......}CH3}\\
\phantom{...}|\\
\ce{CH3 - C - Cl}\\
\phantom{...}|\\
\ce{\phantom{......}CH3}\\
\end{array}\]
Give the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{CH3 - CH - CH2Cl}\\
\phantom{}|\phantom{....}\\
\ce{C2H5}\\
\end{array}\]
Give the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{\phantom{...}CH3}\\
|\\
\ce{\phantom{...}CH2}\\
|\\
\ce{CH2 = CH - CH - C = CH2}\\
\phantom{.............}|\\
\ce{\phantom{.............}Cl}\\
\end{array}\]
Give the IUPAC name of the following compound.
(CH3)3C·CH2·CH2Cl
Give the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{CH3\phantom{.......}}\\
|\phantom{..........}\\
\ce{CH3 - C - CH2 - CH2Cl}\\
|\phantom{..........}\\
\ce{CH3\phantom{.......}}\\
\end{array}\]
Give the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{\phantom{.......................}CH3\phantom{.}}\\
\phantom{....................}|\\
\ce{CH3 - CH2 - CH - CH - C - CH3}\\
\phantom{........}|\phantom{......}|\phantom{......}|\phantom{.}\\
\ce{\phantom{..........}Br\phantom{....}CH3\phantom{..}CH3\phantom{}}\\
\end{array}\]
Give the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{CH3 - CO - CH - CH2 - CH2Cl}\\
|\phantom{.....}\\
\ce{CH3\phantom{..}}
\end{array}\]
Give the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{CH3 - CO - CH - CH2 - CH2Cl}\\
|\phantom{.....}\\
\ce{C2H5\phantom{.}}
\end{array}\]
Write the structure of the following compound and identify it as a 1°, 2° or 3° halide.
2-chloro-2-methylbutane
Write the structure of the following compound and identify it as a 1°, 2° or 3° halide.
2-iodobutane
Write the structure of the following compound and identify it as a 1°, 2°, or 3° halide.
1-bromo-2-methylpropane
Write the structure of the following compound and identify it as a 1°, 2° or 3° halide.
1-chloro-2, 2-dimethylpropane
Write the structure of the following compound and identify it as a 1°, 2° or 3° halide.
chloroethane
Write all the possible chain and position isomers of the compounds having formula C5H11Cl. Write their IUPAC names and identify them as 1°, 2° and 3° halides.
Name the following compound in the IUPAC system:
\[\begin{array}{cc}
\ce{CH3 - CH - CH2 - CH2Br}\\
|\phantom{............}\\
\ce{Cl\phantom{...........}}
\end{array}\]
Name the following compound in the IUPAC system:
\[\begin{array}{cc}
\ce{CH3 - CH - Cl}\\
|\\
\phantom{..}\ce{OH}
\end{array}\]
Name the following compound in the IUPAC system:
\[\begin{array}{cc}
\ce{CH2Cl}\\
|\phantom{.....}\\
\ce{CH2Cl}
\end{array}\]
Name the following compound in the IUPAC system:
CH3CHCl2
Name the following compound in the IUPAC system:
CHCl3
Name the following compound in the IUPAC system:
CCl4
Write the names and possible structures for the compounds having molecular formula, C3H7Cl.
Write the structural formula and IUPAC names of two optically active halides containing five carbon atoms each in their molecules.
How is bromoethane prepared from ethane?
How is bromoethane prepared from ethanol?
How is bromoethane prepared from ethene?
How will you prepare iodoethane from bromoethane?
How will you prepare iodoethane from bromoethane?
How will you prepare bromopropane from butanoic acid?
How will you prepare chloroethane from ethanol?
How will you prepare 1-bromopropane from propene?
Explain the following briefly:
Although haloalkanes are polar in character, yet they are insoluble in water.
Explain the following briefly:
The boiling point of bromoethane is higher than that of chloroethane.
Explain the following briefly:
t-butyl bromide has a lower boiling point than n-butyl bromide.
Explain the following briefly:
Alkyl iodides usually become brown on standing.
Why do alkyl halides show nucleophilic substitution reactions?
Rearrange the following in the order of increasing ease of dehydrohalogenation:
CH3CH2Cl, CH3CHClCH3 and CH3CCl(CH3)2.
Give reasons.
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
Arrange the following in the order of their increasing reactivity in nucleophilic substitution reactions.
CH3F, CH3I, CH3Br, CH3Cl
How will you convert methyl bromide (bromomethane) to acetic acid (ethanoic acid)?
How will you convert prop-1-ene to 1-bromopropane?
How will you convert propan-2-ol to 1-bromopropane?
How will you convert n-propyl bromide to isopropyl bromide?
How will you convert bromoethane to ethoxyethane?
How will you convert propan-1-ol to 2-bromopropane?
Explain why do haloalkanes give alkyl cyanides when treated with KCN but give alkyl isocyanides with silver cyanide?
What is peroxide effect?
Explain with a suitable example of the peroxide effect.
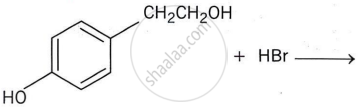
What happens when propene is treated with hydrobromic acid?
What happens when ethanol is treated with thionyl chloride?
What happens when ethanol is treated with phosphorus tribromide?
What happens when bromoethane is treated with alcoholic silver cyanide?
What happens when 1-chloropropane is heated at 573 K?
Which isomer of C4H9Cl will have the lowest boiling point and why?
Complete the following reaction.
\[\ce{CH3 - CH = CH2 + HBr ->[Peroxide]}\]
Complete the following reaction.
\[\ce{CH3CH2CH2Cl ->[Alcoholic KOH][Δ]}\]
Complete the following reaction.
\[\ce{C2H5Cl + Na ->}\]
Complete the following reaction.
\[\ce{CH3Br + KCN -> A ->[H2O/H^+][] B}\]
Complete the following reaction.
\[\ce{(CH3)2CHBr + (alc.)KOH ->[Δ][]}\]
Which is most reactive towards SN1 reaction?
CH3CH2Br, (CH3)2CHBr, (CH3)3C.Br
Which of the following is most reactive for its reaction with HCI?
CH3CH2CH2CH2OH, \[\begin{array}{cc}
\phantom{.........}\ce{OH}\\
\phantom{........}|\\
\ce{CH3 - CH2 - CH - CH3}
\end{array}\], (CH3)2CH-CH2OH, (CH3)3C.OH
Which one among the following pairs does have higher boiling point?
1-bromopropane or 1-iodopropane
Which one among the following pairs does have higher boiling point?
1-bromopropane or 1-bromobutane
Which one among the following pairs does have higher boiling point?
isobutyl bromide or t-butyl bromidec
Identify the compound X, Y and Z in the following sequence of reaction.
\[\ce{C2H5Br ->[alc. KOH][]X->[Br2][]Y->[alc. KOH][]Z}\]
Identify the compound X, Y and Z in the following sequence of reaction.
\[\ce{CH2 = CH2 ->[HBr][]X->[KCN][]Y->[H2O][Dil.acid]Z}\]
Identify the compound X, Y and Z in the following sequence of reaction.
\[\ce{CH3OH ->[HBr][]X->[H2/Ni][525]Y->[Cl2][hv]Z}\]
What is Saytzeff’s rule? Explain with an example.
What are polyhalogen compounds?
Write the structures of three commercially important polyhalogen compounds.
How will you prepare chloroform from acetone (give equations)?
How will you prepare chloroform from ethanol (give equations)?
How will you prepare chloroform from acetone (give equations)?
How is iodoform prepared from acetone?
Show with the help of chemical equations, what happens when chloroform is exposed to sunlight and air for a long time?
Chloroform is a chlorine compound, but it does not give white precipitate with silver nitrate solution. Give reasons.
What happens when chloroform is treated with phenol in the presence of alcoholic KOH at 340 K?
What happens when chloroform is boiled with aqueous KOH?
What happens when chloroform is heated with silver powder?
What happens when reaction:
Aniline reacts with chloroform in the presence of alcoholic potassium hydroxide
What precautions are necessary to be taken for the safe storage of chloroform?
How would you distinguish the following pair of compounds?
Methanol and Ethanol
How would you distinguish the following pair of compounds?
n-propyl alcohol and iso-propyl alcohol
What happens when iodoform is heated with caustic potash?
What happens when chloroform is treated with acetone?
What happens when carbon tetrachloride is heated with dry hydrogen fluoride in the presence of antimony pentachloride?
Explain the following:
The use of carbon tetrachloride as fire extinguisher is not very safe.
Explain the following:
Chloroform is not used as an anaesthetic nowadays.
Explain the following:
A small amount of ethanol should be added to chloroform before its packaging.
Complete the following reaction:
\[\ce{C2H5NH2 + CHCl3 + KOH (alc{.}) ->[Warm]}\]
Complete the following reaction.
\[\ce{CHCl3 + HNO3 ->[\Delta]}\]
Complete the following reaction:
\[\ce{CHI3 + C2H5NH2 + KOH(alc.) ->}\]
Complete the following reaction:
\[\ce{CHCl3 + Cl2 ->[hv][]}\]
Complete the following reaction:
\[\ce{CHCl3 + Ag ->[Heat][]}\]
How will you bring the following conversion?
Iodoform to acetylene
How would you convert carbon tetrachloride to chloroform?
How will you prepare chloroform from acetone (give equations)?
How would you convert chloroform to diethyl carbonate?
How would you convert propanone to iodoform?
What is the iodoform test?
How is the iodoform test carried out?
What is iodoform test significance in organic chemistry?
Write the reactions involved in the preparation of iodoform from propan-2-ol.
What are Freons?
How is feron prepared?
Starting from chloroform how would you prepare acetylene?
Starting from chloroform how would you prepare propyne?
Starting from chloroform how would you prepare chloropicrin?
A sweet smelling organic compound (A) is slowly oxidised by air in the presence of light to a highly poisonous gas. On warming with silver powder, it forms a gaseous substance (B) which is also formed by the action of calcium carbide on water. Identify (A) and (B), and write the chemical equations of the reactions involved.
How is pure chloroform prepared?
How is pure chloroform stored?
How is ethyl bromide prepared in the laboratory?
Describe ethyl bromide important synthetic applications.
Write the IUPAC name of the following compound.
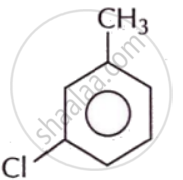
Write the IUPAC name of the following compound.
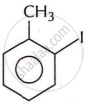
Write the IUPAC name of the following compound.

Write the IUPAC name of the following compound.
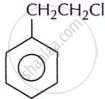
Write the IUPAC name of the following compound.
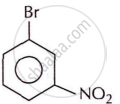
Write the IUPAC name of the following compound.
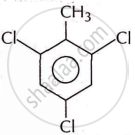
Write the structural formula and give the IUPAC name of the following:
o-bromotoluene
Write the structural formula and give the IUPAC name of the following:
Benzyl chloride
Write the structural formula and give the IUPAC name of the following:
Benzotrichloride
Write the structural formula and give the IUPAC name of the following:
o-chlorobenzene sulphonic acid
Describe a method for the preparation of haloarenes from diazonium salts.
Describe a method for the preparation of haloarenes from benzene.
How many isomers are possible for the compound C7H7Cl? Write their structures and give their IUPAC names.
Why do alkyl halides (haloalkanes) undergo hydrolysis more easily than aryl halides (haloarenes)?
Account for the fact that halogen in chlorobenzene is less reactive than in methyl chloride.
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
Arrange the following compounds in the order of increasing reactivity towards nucleophilic substitution reactions:
- Chlorobenzene
- 2, 4-dinitrochlorobenzene
- 4-nitrochlorobenzene
- 2, 4, 6-trinitrochlorobenzene
Explain the following briefly.
In chlorobenzene, the electrophilic substitution takes place at o- and p-positions.
Explain the following briefly.
Allyl chloride is hydrolysed readily as compared to 1-chloropropane.
Explain the following briefly.
Vinyl chloride is less reactive than ethyl chloride.
Explain the following briefly.
The electrophilic substitution reactions in haloarenes occur slowly as compared to those in benzene.
What happens when chlorobenzene is treated with ethyl chloride in the presence of sodium in dry ether?
What happens when chlorobenzene is heated with aqueous ammonia in the presence of cuprous oxide at 475 K and under high pressure?
What happens when iodobenzene is heated with copper powder in a sealed tube?
What happens when benzene diazonium chloride is treated with an aqueous solution of potassium iodide?
What happens when chlorine is passed in boiling toluene in the presence of sunlight?
Write the resonance structures of chlorobenzene and explain.
Why is chlorobenzene less reactive than chloroethane towards nucleophiles?
Explain whether chloroethene should be more or less reactive than chloroethane towards nucleophiles.
Account for the following:
Haloalkanes are more reactive than haloarenes.
Account for the following:
Haloalkanes undergo nucleophilic substitution, whereas haloarenes undergo electrophilic substitution.
Give the structure and name of the product in the following reaction.
Chlorination of toluene in the presence of anhydrous AlCl3.
Give the structure and name of the product in the following reaction.
Sulphonation of chlorobenzene.
Give the structure and name of the product in the following reaction.
Nitration of bromobenzene.
Give the structure and name of the product in the following reaction.
Friedel-Crafts methylation of chlorobenzene.
How will you distinguish C2H5Br from C6H4Br?
What is Sandmeyer’s reaction?
Illustrate with a suitable example of Sandmeyer’s reaction?
How is the following conversion carried out?
Chlorobenzene to benzoic acid
How is the following conversion carried out?
Chlorobenzene to benzene
How the following conversion can be carried out?
Aniline to chlorobenzene
How can the following conversion be brought about?
Chlorobenzene to phenol
How is the following conversion carried out?
Chlorobenzene to benzylamine
How is the following conversion carried out?
Chlorobenzene to aniline.
Complete the following chemical equation.
\[\ce{C6H5N2Cl + HCl ->[CuCl][]?}\]
Complete the following chemical equation.
\[\ce{C6H5N2Cl + Conc{.}HNO3 ->[H2SO4(Conc{.})][]?}\]
Complete the following chemical equation.
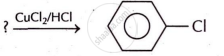
Complete the following chemical equation.
\[\begin{array}{cc}
\ce{\phantom{.....}O}\\
\phantom{.....}||\\
\ce{C6H5Cl + CH3-C-Cl ->[AlCl3][]?}
\end{array}\]
Complete the following chemical equation.
\[\ce{C6H5Br + 2Na + CH3Br ->[dry ether][]?}\]
Complete the following chemical equation.
\[\ce{2C6H5Cl + 2Na ->[Dry ether][]?}\]
Identify X, Y and Z in the following sequence of reactions:
\[\ce{C6H6 ->[Cl2,FeCl3][]X->[NaOH_{(aq)}][263K, 300atm] Y ->[Dil{.}HCl][]Z}\]
Identify X, Y and Z in the following sequence of reactions:
\[\ce{C5H5NH2 ->[Na NO2, HCl][273K]X->[CuBr, HBr][]Y->[CH3Cl][AlCl3]Z}\]
Identify X, Y and Z in the following sequence of reactions:
\[\ce{C6H5Cl->[CuCl, HCl][]X->[CuCn][Pyridine, Δ]Y->[Dil{.}HCl][]Z}\]
How will you distinguish between the following pair of compounds?
Chlorobenzene and benzyl chloride
How will you distinguish between the following pair of compounds?
o-chlorotoluene and benzyl chloride
How will you distinguish between the following pair of compounds?
Bromobenzene and benzyl bromide
How will you distinguish C2H5Br from C6H4Br?
Give one example of the following reaction:
Wurtz Reaction
Give one example of the following reaction:
Wurtz-Fittig Reaction
Nootan solutions for Chemistry [English] Class 12 ISC 10 Haloalkanes and Haloarenes VERY SHORT ANSWER TYPE QUESTIONS [Pages 608 - 609]
Give one example of a 1° haloalkane.
Give one example of a 2° haloalkane.
Give an example of a 3° alkyl halide.
Which of the following are nuclear halogen derivatives?

Give one example of a primary alcohol.
Give one example of a secondary alcohol.
Give one example of tertiary alcohol.
Write the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{Br\phantom{...}Br\phantom{.......}}\\
|\phantom{.....}|\phantom{........}\\
\ce{CH3-CH-CH-COOC2H5}
\end{array}\]
Write the IUPAC name of the following compound.
(CH3)3C.CH2.CH2Cl
Write the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{\phantom{...................}CH3}\\
\phantom{.................}|\\
\ce{CH3-CH2-CH-CH-C-CH3}\\
\phantom{.......}|\phantom{.....}|\phantom{.....}|\phantom{.}\\
\ce{\phantom{.........}Br\phantom{...}CH3\phantom{..}CH3\phantom{.}}\\
\end{array}\]
Write the IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{\phantom{...........}CH3}\\
\phantom{.........}|\\
\ce{CH3-CH2-CH-C-CH2Cl}\\
\phantom{....}|\phantom{.....}|\\
\ce{\phantom{......}Br\phantom{...}CH3}
\end{array}\]
Write the IUPAC name of the following:
n-butyl bromide
Write the IUPAC name of the following:
tert-butyl bromide
Write the IUPAC name of the following:
sec-pentyl bromide
Write the position isomers of C4H9Br.
What is the order of the ease of replacement of different types of H atoms by halogen atoms in alkanes?
Among PCl5 and SOCl5, which reagent is preferred for the preparation of chloroalkanes from alcohols?
Arrange 1-bromobutane, 1-bromo-2-methylpropane and 2-bromo-2-methylpropane in the order of their increasing boiling points.
Inspite of being polar, why are haloalkanes insoluble in water?
Why do alkyl iodides become brown or violet on standing?
Arrange 1°, 2° and 3° haloalkanes in the decreasing order of their reactivity towards nucleophilic substitution reactions.
Name the reagent used to convert a haloalkane into an alcohol.
Name the reagent used to convert a haloalkane into an ether.
Name the reagent used to convert a haloalkane into an amine.
Name the reagent used to convert a haloalkane into an alkene.
Arrange primary, secondary and tertiary haloalkanes in the decreasing order of the ease of dehydrohalogenation.
What happens when an alkyl iodide is heated with hydroiodic acid in the presence of red phosphorus?
How will you convert ethyl bromide into ethyl alcohol?
How will you convert methyl bromide (bromomethane) to acetic acid (ethanoic acid)?
How will you convert ethyl bromide into diethyl ether?
What are arenes?
Give two examples of arenes.
Sort out o-, p- and m-directing groups among the following:
-OH, -NO2, -CONH2, -CN, -Cl, -CH3, -COOH, -OCH3
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
How can the following conversion be brought about?
Chlorobenzene to phenol
How is the following conversion carried out?
Chlorobenzene to aniline.
How is the following conversion carried out?
Chlorobenzene to benzoic acid
How is chlorobenzene converted into diphenyl/biphenyl?
How is chlorobenzene converted into diphenyl/biphenyl?
How would you obtain pure chloroform in the laboratory?
Why is chloroform stored in brown bottles?
What happens when chloroform is subjected to nitration?
What happens when chloroform is subjected to hydrolysis?
What happens when chloroform is subjected to dehalogenation?
Which of the following pair of compounds can be distinguished by iodoform test?
Methanol and ethanol
Which of the following pair of compounds can be distinguished by iodoform test?
Methanol and butan-1-ol
Which of the following pair of compounds can be distinguished by iodoform test?
Butan-1-ol and butan-2-ol
What is pyrene?
What is pyrene used for?
Draw structure of DDT.
Write the IUPAC name of the following:
DDT
Write the structure of the following:
Gammaxene
Write the IUPAC name of the following:
Gammaxene
What happens when (write chemical equations only) silver acetate is heated with bromine in the presence of CCl4?
What happens when (write chemical equations only) n-propyl bromide is treated with sodium ethoxide?
What happens when (write chemical equations only) iso-propyl chloride is treated with silver nitrite?
What happens when (write chemical equations only) benzene is heated with iodine in the presence of iodic acid?
What happens when (write chemical equations only) benzenediazonium chloride is heated with hydrobromic acid in the presence of cuprous bromide?
Complete the following chemical equation
\[\ce{CHCl3 + C2H5NH2 + 3KOH -> {.....}}\]
How the following conversion can be carried out?
1-Bromopropane to 2-bromopropane
How would you convert propanone to iodoform?
Give the IUPAC name of the following compound:
CH3CH(Cl)CH(Br)CH3
Give the IUPAC name of the following compound:
CHF2CBrClF
Give the IUPAC name of the following compound:
ClCH2C≡CCH2Br
Give the IUPAC name of the following compound:
(CCl3)3CCl
Give the IUPAC name of the following compound:
CH3C(p-ClC6H4)2CH(Br)CH3
Give the IUPAC name of the following compound:
(CH3)3CCH=CClC6H4I-p
Give the IUPAC name of the following compound:
CH3CHCl(CH2)2CCl2C2H5
Which compound in the following pair will react faster in SN2 reaction with OH−?
CH3Br or CH3I
Which compound in the following pair will react faster in SN2 reaction with OH−?
(CH3)3CCl or CH3Cl
Which compound in the following pair will react faster in SN2 reaction with OH−?
CH2=CHBr or CH2=CH-CH2Br
Arrange the following compounds in increasing order of SN1 reactivity.
ClCH2CH = CHCH2CH3, CH3C(Cl) = CHCH2CH3, CH3CH = CHCH2CH2Cl.
Arrange the following compounds in increasing order of SN1 reactivity.
CH3CH2Br, CH2 = CHCH(Br)CH3, CH2 = CHBr, CH3CH(Br)CH3.
Arrange the following compounds in increasing order of SN1 reactivity.
(CH3)CCl, C6H5C(CH3)2Cl, (CH3)2CHCl, CH3CH2CH2Cl.
Predict the order of reactivity of the following compounds in dehydrohalogenation:
CH3CH2CH2CH2Cl, (CH3)2CHCH2Cl, (CH3)2CH-CH2Br, CH3CH(Br)CH2CH3, (CH3)3C-Br
Predict the order of reactivity of the following compounds in dehydrohalogenation:
CH3CH(Br)CH3, CH3CH2CH2Br, (CH3)2CH-CH2Br, (CH3)3C-CH2Br
Which of the following compounds will give positive iodoform test?
Butan-1-ol, Butan-2-ol, Tert-butyl alcohol, Ethanol, Propanol, Propanone, Butan-2-one, Pentan-3-one, Cyclohexanone, 1-methylcyclohexanol, 1-phenylethanol, 2- phenylethanol?
Nootan solutions for Chemistry [English] Class 12 ISC 10 Haloalkanes and Haloarenes SHORT ANSWER TYPE QUESTIONS [Pages 611 - 612]
How are haloalkanes classified?
Among aromatic halogen compounds, how are nuclear derivatives different from side chain derivatives? Explain with examples.
How is bromoethane prepared from ethene?
How is bromoethane prepared from ethanol?
How would you obtain bromoethane from silver propanoate?
Explain with an example of the following reaction:
Finkelstein reaction
Explain with an example of the following reaction:
Hunsdiecker reaction
Why are the melting points and boiling points of haloalkanes much higher than those of the parent alkanes?
Why do the boiling points of isomeric haloalkanes decrease with increase in branching?
Explain the following briefly:
Although haloalkanes are polar in character, yet they are insoluble in water.
Why are haloalkanes more reactive than haloarenes?
Why do 3° alkyl halides undergo substitution by SN1 mechanism, whereas 1° alkyl halides by SN2 mechanism?
How will you carry out the following conversion?
2-bromopropane to propan-2-ol
How will you carry out the following conversion?
Bromoethane to methoxyethane.
How will you carry out the following conversion?
Iodomethane to dimethylamine
How will you carry out the following conversion?
1-bromobutane to but-1-ene
How will you carry out the following conversions?
1-chloropropane to 2-chloropropane
What is an ambident group?
Why does potassium cyanide give a nitrile while silver cyanide an isonitrile when treated with an alkyl halide?
What is Saytzeff’s rule? Explain with an example.
What are haloarenes?
Give two examples of haloarenes.
How will you obtain chlorobenzene from benzene?
How will you obtain iodobenzene from benzene?
How will you obtain bromobenzene from benzene diazonium chloride?
How the following conversion can be carried out?
Aniline to chlorobenzene
Discuss the mechanism of nuclear halogenation of benzene and explain the role of halogen carrier.
Why do isomeric (o-, m- and p-) dihalobenzenes possess almost similar boiling points?
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Explain the low reactivity of haloarenes as compared to haloalkanes on the basis of resonance.
Why is the C-Cl bond length in chlorobenzene shorter as compared to that in chloromethane?
Why is the C-Cl bond in chlorobenzene less polar as compared to that in chloromethane?
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
How can the following conversion be brought about?
Chlorobenzene to phenol
How is the following conversion carried out?
Chlorobenzene to aniline.
How would you convert chlorobenzene to p-chloroacetophenone?
How is chlorobenzene converted into diphenyl/biphenyl?
How would you convert chlorobenzene to toluene?
How is the following conversion carried out?
Chlorobenzene to benzene
Among chlorobenzene and 4-nitrochlorobenzene, which is more reactive towards nucleophilic substitution reactions and why?
Why do haloarenes undergo electrophilic substitutions at o- and p-positions?
During electrophilic substitution reactions of haloarenes, the p-isomer usually dominates. Explain, why?
How will you prepare chloroform from ethanol (give equations)?
How would you obtain chloroform from propanone?
Why is it necessary to take extra precautions for the safe storage of chloroform and what are the precautions?
What happens when chloroform is treated with zinc and hydrochloric acid?
What happens when chloroform is boiled with aqueous KOH?
How will you bring the following conversion?
Iodoform to acetylene
Write a chemical reaction useful to prepare the following:
Carbon tetrachloride from carbon disulphide.
What happens when carbon tetrachloride is treated with steam at high temperatures?
What happens when carbon tetrachloride is treated with moist iron filings?
What is carbylamine reaction?
What is carbylamine reaction significance?
How is iodoform prepared from acetone?
What is the iodoform test?
What type of compounds respond to the iodoform test?
Give a simple chemical test to distinguish between the following pair of compounds:
Pentan-2-one and Pentan-3-one
What are Freons?
How is feron prepared?
Discuss uses of freons.
What is DDT?
Explain the preparation of the following compound.
DDT
Write the structure of the following:
Gammaxene
Write the preparation of benzene hexachloride.
Write the uses of benzene hexachloride.
Account for the following:
Haloalkanes are more reactive than haloarenes.
Account for the following:
Haloalkanes undergo nucleophilic substitution, whereas haloarenes undergo electrophilic substitution.
Describe with chemical equation what happens when chloromethane is reacted with toluene in the presence of anhydrous aluminium chloride?
Account for the following:
Haloarenes are insoluble in water but are soluble in benzene.
Account for the following:
Haloalkanes are more reactive than haloarenes.
Account for the following:
Haloalkanes undergo nucleophilic substitution reactions.
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)2CHCH(Cl)CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH2CH(CH3)CH(C2H5)Cl
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH2C(CH3)2CH2I
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)3CCH2CH(Br)C6H5
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH(CH3)CH(Br)CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(C2H5)2CH2Br
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH = C(Cl)CH2CH(CH3)2
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH = CHC(Br)(CH3)2
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
p-ClC6H4CH2CH(CH3)2
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
m-ClCH2C6H4CH2C(CH3)3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
o-Br-C6H4CH(CH3)CH2CH3
Write the structure of the following compound:
2-Chloro-3-methylpentane
Write the structure of the following organic halogen compound.
p-Bromochlorobenzene
Write the structure of the following compound:
1-Chloro-4-ethylcyclohexane
Write the structure of the following organic halogen compound.
2-(2-Chlorophenyl)-1-iodooctane
Write the structure of the following organic halogen compound:
Perfluorobenzene
Write the structure of the following compound:
4-tert. Butyl-3-iodoheptane
Write the structure of the following compound:
1-Bromo-4-sec. butyl-2-methylbenzene
Write the structure of the following compound:
1,4-Dibromobut-2-ene
A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
A hydrocarbon C5H12 gives only one monochlorination product. Identify the hydrocarbon.
What are ambident nucleophiles? Explain with an example.
Write the equation for the preparation of 1-iodobutane from 1-butanol.
Write the equation for the preparation of 1-iodobutane from 1-chlorobutane.
Write the equation for the preparation of 1-iodobutane from but-1-ene.
How would you prepare following compound using a nucleophilic substitution reaction? Mention the equations only.
CH3OC(CH3)2
How would you prepare following compound using a nucleophilic substitution reaction? Mention the equations only.
CH3C ≡ CCH2CH3
How would you prepare following compound using a nucleophilic substitution reaction? Mention the equations only.
CH3CH2CH2N(CH3)2
How would you prepare following compound using a nucleophilic substitution reaction? Mention the equations only.
C6H5 - CH2OCOCH3
How would you prepare following compound using a nucleophilic substitution reaction? Mention the equations only.
CH3CH2CH2CH2NO2
How would you prepare following compound using a nucleophilic substitution reaction? Mention the equations only.
CH3CH2CH2CN
How would you prepare following compound using a nucleophilic substitution reaction? Mention the equations only.
C6H5CH2N+ ≡ C−
How would you prepare following compound using a nucleophilic substitution reaction? Mention the equations only.
CH3CH2 − O − N = O
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
1-Bromo-1-methylcyclohexane
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
Cyclohexylmethylbromide
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
2-Chloro-2-methylbutane
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene.
3-bromopent-1-ene
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
2, 2, 3-Trimethyl-3-bromopentane
The nucleophilic substitution of primary alkyl chlorides with sodium acetate is catalysed by sodium iodide. Explain why.
In the following reaction, a new ring is formed. Use curved arrow notation to explain the formation of the new ring.
1, 4-dibromobutane (0.1 mol) is treated with sodium sulphide (0.1 mol) in aqueous ethanol.
In the following reaction, a new ring is formed. Use curved arrow notation to explain the formation of the new ring.
1-chloro-2-(2-hydroxyphenyl) ethane is treated with aqueous sodium hydroxide.
Why does p-methoxybenzyl bromide react faster than p-nitrobenzyl bromide with ethanol to form an ether product?
Explain the formation of the product in the following reaction:
\[\ce{CH3CH = CHCH2Cl + H2O->CH3CH = CHCH2OH + CH3CH(OH)CH = CH2}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2Cl + NaI ->[acetone][heat]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{(CH3)3CBr + KOH ->[ethanol][heat]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq.ethanol]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{(CH3)3 CBr + H2O ->[Heat][]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{(CH3)2CH - CH(Br) CH2CH3->[C2H5ONa][Ethanol/Heat]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Cl + SbF3->[Heat]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH2 = CHCH2Br + CH3C ≡ CNa ->[Liq{.} NH3]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH = C(CH3)2 + HBr ->}\]
Write the structure of the major organic product in the following reaction:
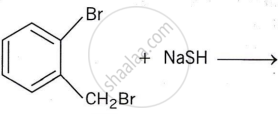
Write the structure of the major organic product in the following reaction:
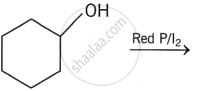
Write the structure of the major organic product in the following reaction:
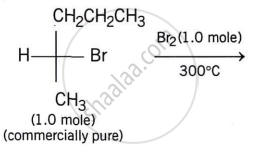
In the following monobromination reaction, the number of possible chiral products is
Nootan solutions for Chemistry [English] Class 12 ISC 10 Haloalkanes and Haloarenes LONG ANSWER TYPE QUESTIONS [Page 615]
What are haloalkanes?
What are haloarenes?
How are haloalkanes classified?
How are haloarenes classified? Give suitable examples.
Write short notes on Markownikoff ’s rule.
Explain with a suitable example of the peroxide effect.
Write a short note on the allylic substitution.
Mention any three methods of preparation of haloalkanes from alcohols.
What is halide exchange method for the preparation of iodoalkanes?
Discuss the relative stability of different types of haloalkanes.
Why do alkyl iodides become brown or violet on standing?
Why do alkyl halides show nucleophilic substitution reactions?
Discuss the mechanisms of SN1 and SN2 reactions shown by nucleophilic substitution reactions.
Explain why do haloalkanes give alkyl cyanides when treated with KCN but give alkyl isocyanides with silver cyanide?
Explain why:
Alkyl halides form nitroalkanes on treatment with silver nitrite but form alkyl nitrites on treatment with potassium nitrite.
What is Saytzeff’s rule? Explain with an example.
On the basis of Saytzeff’s rule, explain the ease of dehydrohalogenation of different types of haloalkanes.
Discuss some important methods of preparation of haloarenes.
How are aralalkyl halides prepared?
Explain the lower reactivity of haloarenes as compared to haloalkanes on the basis of hybridisation state of carbon of C−X bond.
What is the effect of substituents on the reactivity of haloarenes? Explain with suitable examples.
Explain why the electrophilic substitution reactions in haloarenes occur slowly and require more drastic conditions as compared to those in benzene.
Explain why:
Haloarenes undergo electrophilic substitution at o- and p-positions and not at m-positions.
Write a short note on Fittig reaction.
Write a short note on Wurtz-Fittig reaction.
Write a short note on Ullmann reaction.
How is iodoform prepared from ethanol? Give balanced equation.
How is iodoform prepared from acetone?
What is the iodoform test?
How is the iodoform test carried out?
Discuss some important applications of the iodoform test.
What are polyhalogen compounds?
Explain the preparation of the following compound.
DDT
Write the preparation of benzene hexachloride.
How is feron prepared?
Give the uses of DDT.
Write the uses of benzene hexachloride.
Discuss uses of freons.
How will you bring about the following conversion in not more than two steps?
Ethanol to But-1-yne
How will you bring about the following conversion in not more than two steps?
Ethene to Bromoethene
How will you bring about the following conversion?
Propene to 1-nitropropane
How will you bring about the following conversion?
Toluene to benzyl alcohol
How will you bring about the following conversion?
Propene to propyne
How will you bring about the following conversion?
Ethanol to ethyl fluoride
How will you bring about the following conversion?
Bromomethane to propanone
How will you bring about the following conversion?
But-1-ene to but-2-ene
How will you bring about the following conversion?
1-Chlorobutane to n-octane
How will you bring about the following conversion in not more than two steps?
Bromoethane to cis-Hex-3-ene
How will you bring about the following conversion in not more than two steps?
Benzyl alcohol to Phenylethanenitrile
How are the following conversions carried out?
benzene to biphenyl
How will you bring about the following conversion in not more than two steps?
Cyclopentene to Cyclopenta-1, 3-diene
How will you bring about the following conversion in not more than two steps?
Aniline to Phenyl isocyanide
How would you distinguish the following pair of compounds?
Methanol and Ethanol
How will you distinguish between propan-1-ol and propan-2-ol?
How will you distinguish between the following pair of compounds?
Chlorobenzene and benzyl chloride
How will you distinguish between n-Butylamine and diethylamine?
Give reasons:
The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Explain why alkyl halides, though polar, are immiscible with water?
Explain why vinyl chloride is unreactive in nucleophilic substitution reactions.
Explain why neopentylbromide undergoes nucleophilic substitution reactions very slowly.
Explain why 3-bromocyclohexene is more reactive than 4-bromocyclohexene in hydrolysis with aqueous NaOH.
Explain why tert-butyl bromide reacts with aqueous sodium hydroxide by SNl mechanism while n-butyl chloride reacts by SN2 mechanism.
Explain why Grignard reagents should be prepared under anhydrous conditions?
Nootan solutions for Chemistry [English] Class 12 ISC 10 Haloalkanes and Haloarenes OBJECTIVE (MULTIPLE CHOICE) TYPE QUESTIONS [Pages 616 - 617]
The decreasing order of reactivity of alkyl halides is ______.
RI > RCl > RBr
RBr > RCl > RI
RI > RBr > RCl
Cl > RBr > Rl
A sample of chloroform, before using as an anaesthetic, is tested ______.
by Fehling solution
by ammonical cuprous chloride
by ammonical silver nitrate
after boiling with alc. KOH, with silver nitrate
In the reaction \[\ce{Primary amine + CHCl3 + alc{.} KOH -> Products}\], the main product is ______.
cyanide
isocyanide
acid
aldehyde
When propene is heated at 400°C in the presence of chlorine, it gives ______.
polyvinyl chloride
no reaction
1, 2-dichloropropane
allyl chloride
Which process does not occur during the formation of CHCl3 from C2H5OH and bleaching powder?
Hydrolysis
Oxidation
Elimination
Chlorination
Chlorine reacts with ethanol to give ______.
diethyl chloride
chloroform
acetaldehyde
chloral
The compound with the highest boiling point is ______.
CH4
CH3OH
CH3Cl
CH3OCH3
Haloform reaction does not take place with ______.
acetone
2-chloropropane
ethanol
methanol
The number of possible isomers for the compound C2H3Cl2Br is ______.
2
3
4
5
Which of the following is a 2° alkyl halide?
CH3CH2Br
\[\begin{array}{cc}
\ce{Cl\phantom{.........}}\\
|\phantom{.........}\\
\ce{CH3 - CH - CH2CH3}
\end{array}\](CH3)3C.Cl
CH3Br
The IUPAC name of sec-butyl bromide is ______.
1-bromobutane
2-bromobutane
1-bromo-2-methylpropane
2-bromo-2-methylpropane
Nootan solutions for Chemistry [English] Class 12 ISC 10 Haloalkanes and Haloarenes FILL IN THE BLANKS TYPE QUESTIONS
Solutions for 10: Haloalkanes and Haloarenes
![Nootan solutions for Chemistry [English] Class 12 ISC chapter 10 - Haloalkanes and Haloarenes Nootan solutions for Chemistry [English] Class 12 ISC chapter 10 - Haloalkanes and Haloarenes - Shaalaa.com](/images/chemistry-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Nootan solutions for Chemistry [English] Class 12 ISC chapter 10 - Haloalkanes and Haloarenes
Shaalaa.com has the CISCE Mathematics Chemistry [English] Class 12 ISC CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Nootan solutions for Mathematics Chemistry [English] Class 12 ISC CISCE 10 (Haloalkanes and Haloarenes) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Nootan textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry [English] Class 12 ISC chapter 10 Haloalkanes and Haloarenes are Overview of Haloalkanes and Haloarenes.
Using Nootan Chemistry [English] Class 12 ISC solutions Haloalkanes and Haloarenes exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Nootan Solutions are essential questions that can be asked in the final exam. Maximum CISCE Chemistry [English] Class 12 ISC students prefer Nootan Textbook Solutions to score more in exams.
Get the free view of Chapter 10, Haloalkanes and Haloarenes Chemistry [English] Class 12 ISC additional questions for Mathematics Chemistry [English] Class 12 ISC CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.
