Advertisements
Advertisements
Question
Give the structure and name of the product in the following reaction.
Nitration of bromobenzene.
Chemical Equations/Structures
Advertisements
Solution
When bromobenzene is treated with concentrated nitric acid and concentrated sulphuric acid, it undergoes electrophilic substitution to form ortho and para nitro derivatives.
- Reaction:
\[\ce{C6H5Br + HNO3 ->[conc. H2SO4, heat][]\underset{o-bromonitrobenzene}{o-BrC6H4NO2} + \underset{p-bromonitrobenzene}{p-BrC6H4NO2}}\] - Structures:
- Ortho-bromonitrobenzene (1-bromo-2-nitrobenzene)
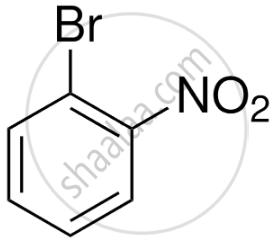
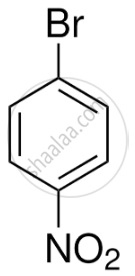
- Para-bromonitrobenzene (1-bromo-4-nitrobenzene)
- Ortho-bromonitrobenzene (1-bromo-2-nitrobenzene)
- Names of the Products:
- Ortho-bromonitrobenzene (Minor product)
- Para-bromonitrobenzene (Major product)
shaalaa.com
Is there an error in this question or solution?
