Advertisements
Advertisements
Questions
Give a simple chemical test to distinguish between the following pair of compounds:
Pentan-2-one and Pentan-3-one
Can you distinguish pentan-2-one and pentan-3-one by iodoform test?
Advertisements
Solution
Pentan-2-one and pentan-3-one can be distinguished by the iodoform test.
Iodoform test: Pentan-2-one is a methyl ketone, so it responds to the iodoform test. However, pentan-3-one is not a methyl ketone and does not respond to this test.
Pentan-2-one has a methyl ketone group (CH3-CO-) at the second carbon. Compounds with a methyl ketone group give a positive iodoform test, producing a yellow precipitate of iodoform (CHI3).
\[\begin{array}{cc}
\ce{O}\phantom{.................................................}\\
||\phantom{.................................................}\\
\ce{\underset{Pentan-2-one}{CH3CH2CH2 - C - CH3} + 3NaOI ->\underset{Sodium butanoate}{CH3CH2CH2COONa} + \underset{(yellow ppt)}{\underset{Iodoform}{CHI3\downarrow}} + 2NaOH}\
\end{array}\]
Pentan-3-one has the carbonyl group at the third position, so it does not have a methyl ketone structure. It does not give a positive iodoform test.
\[\begin{array}{cc}
\ce{O}\phantom{.......................................}\\
||\phantom{.......................................}\\
\ce{\underset{Pentan-3-one}{CH3CH2 - C - CH2CH3} + NaOI -> No yellow ppt of iodoform}\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Distinguish between: CH3COOH and HCOOH
Predict the products of the following reactions :

Propanal and Propanone
Out of CH3CH2 – CO – CH2 – CH3 and CH3CH2 – CH2 – CO – CH3, which gives iodoform test?
How will you convert ethanal into the following compound?
But-2-enoic acid
Give a simple chemical test to distinguish between the following pair of compounds:
Acetophenone and Benzophenone
Give a simple chemical test to distinguish between the following pair of compounds:
Phenol and Benzoic acid
Give a simple chemical test to distinguish between the following pair of compounds:
Benzoic acid and Ethyl benzoate
Complete the synthesis by giving missing starting material, reagent or product.

Which of the following compounds will give butanone on oxidation with alkaline \[\ce{KMnO4}\] solution?
Oxidation of ketones involves carbon-carbon bond cleavage. Name the products formed on oxidation of 2, 5-dimethylhexan-3-one.
Acetone and acetaldehyde are differentiated by
Ammonical silver nitrate solution is called
A hydrocarbon (A) with molecular formula C5H10 on ozonolysis gives two products (B) and (C). Both (B) and (C) give a yellow precipitate when heated with iodine in presence of NaOH while only (B) give a silver mirror on reaction with Tollen’s reagent.
- Identify (A), (B) and (C).
- Write the reaction of B with Tollen’s reagent.
- Write the equation for iodoform test for C.
- Write down the equation for aldol condensation reaction of B and C.

Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
The correct set of products obtained in the following reactions:
- \[\ce{RCN ->[reduction]}\]
- \[\ce{RCN ->[(i) CH3MgBr][(ii) H2O]}\]
- \[\ce{RNC ->[hydrolysis]}\]
- \[\ce{RNH2 ->[HNO2]}\]
You are given four organic compounds “A”, “B” , “C” and “D”. The compounds “A”, “B” and “C” form an orange-red precipitate with 2, 4 DNP reagent. Compounds “A” and “B” reduce Tollen’s reagent while compounds “C” and “D” do not. Both “B” and “C” give a yellow precipitate when heated with iodine in the presence of NaOH. Compound “D” gives brisk effervescence with sodium bicarbonate solution. Identify “A”, “B”, “C” and “D” given the number of carbon atoms in three of these carbon compounds is three while one has two carbon atoms. Give an explanation for our answer.
Match List-I with List-II:
| List-I (Reaction) |
List-II (Reagents/Condition) |
||
| A. | 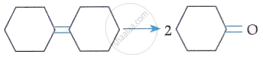 |
I. | 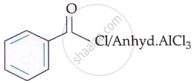 |
| B. | 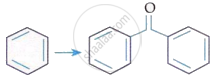 |
II. | CrO3 |
| C. | 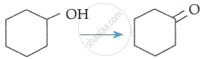 |
III. | KMnO4/KOH, Δ |
| D. |  |
IV. | (i) O3 (ii) Zn-H2O |
Choose the correct answer from the options given below:
