Advertisements
Advertisements
Questions
How will you bring about the following conversion?
Bromomethane to propanone
How will you bring about the following conversion in not more than two steps?
Bromomethane to propanone
Advertisements
Solution 1
\[\begin{array}{cc}
\ce{\underset{Bromomethane}{CH3 - Br} ->[Alc{.} KCN][C2H5OH, -H2O, \Delta] \underset{Acetonitrile}{CH3CN} ->[CH3MgBr/ether] [CH3 - C = NMgBr] ->[H^+/H2O][-NH3, -Mg(OH)Br] CH3 - C = O}\\
\phantom{....................................................................................}|\phantom{.........................................................}|\\
\phantom{.........................................................................................}\ce{CH3}\phantom{................................................}\ce{\underset{propanone}{CH3}}\\
\end{array}\]
Solution 2
\[\begin{array}{cc}
\ce{\underset{Bromomethane}{CH3 - Br} ->[HC = C- Na+][Liq{.} NH3, -NaBr] \underset{Propyne}{CH3 - C ≡ CH} ->[Dil{.} H2SO4][HgSO4] CH3 - C - CH3}\\
\phantom{................................................................................}||\\
\phantom{................................................................................}\ce{\underset{propanone}{O}}\\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
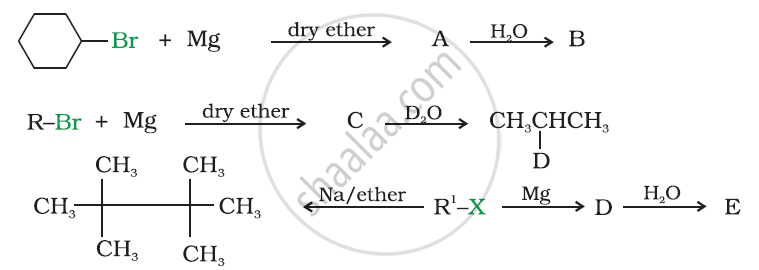
Identify A, B, C, D, E, R and R1 in the following:
What happens when bromobenzene is treated with Mg in the presence of dry ether?
How the following conversion can be carried out?
2-Chlorobutane to 3, 4-dimethylhexane
Write the structure of main compounds A and B in the following reaction:
In the preparation of chlorobenzene from aniline, the most suitable reagent is:
A Grignard reagent may be made by reacting magnesium with ____________.
Which of the following alkyl halides is used as a methylating agent?
Which of the following statements are correct about the reaction intermediate?

(i) Intermediate (c) is unstable because in this carbon is attached to 5 atoms.
(ii) Intermediate (c) is unstable because carbon atom is sp2 hybridised.
(iii) Intermediate (c) is stable because carbon atom is sp2 hybridised.
(iv) Intermediate (c) is less stable than the reactant (b).
Why is it necessary to avoid even traces of moisture during the use of a Grignard reagent?
Reaction of Grignard reagent with aromatic aldehyde and subsequent aqueous treatment produces
 A product (1) of above reaction is:-
A product (1) of above reaction is:-
The product formed in the first step of the reaction
\[\begin{array}{cc}
\ce{Br}\phantom{......}\\
|\phantom{.......}\\
\ce{CH3-CH2-CH-CH2-CH-CH3}\\
\phantom{...............}|\\
\phantom{................}\ce{Br}
\end{array}\]
with excess Mg/Et2O(Et = C2H5) is:
Explain why Grignard reagents should be prepared under anhydrous conditions.
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
