Advertisements
Advertisements
प्रश्न
How will you bring about the following conversion?
Bromomethane to propanone
How will you bring about the following conversion in not more than two steps?
Bromomethane to propanone
Advertisements
उत्तर १
\[\begin{array}{cc}
\ce{\underset{Bromomethane}{CH3 - Br} ->[Alc{.} KCN][C2H5OH, -H2O, \Delta] \underset{Acetonitrile}{CH3CN} ->[CH3MgBr/ether] [CH3 - C = NMgBr] ->[H^+/H2O][-NH3, -Mg(OH)Br] CH3 - C = O}\\
\phantom{....................................................................................}|\phantom{.........................................................}|\\
\phantom{.........................................................................................}\ce{CH3}\phantom{................................................}\ce{\underset{propanone}{CH3}}\\
\end{array}\]
उत्तर २
\[\begin{array}{cc}
\ce{\underset{Bromomethane}{CH3 - Br} ->[HC = C- Na+][Liq{.} NH3, -NaBr] \underset{Propyne}{CH3 - C ≡ CH} ->[Dil{.} H2SO4][HgSO4] CH3 - C - CH3}\\
\phantom{................................................................................}||\\
\phantom{................................................................................}\ce{\underset{propanone}{O}}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
How will you bring about the following conversion?
1-Chlorobutane to n-octane
Explain why Grignard reagents should be prepared under anhydrous conditions?
What happens when methyl bromide is treated with sodium in the presence of dry ether?
In the preparation of chlorobenzene from aniline, the most suitable reagent is:
What would be the reactant and reagent used to obtain 2, 4-dimethyl pentan-3-ol?
Draw other resonance structures related to the following structure and find out whether the functional group present in the molecule is ortho, para directing or meta directing.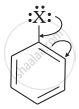
Identify the following named reaction:
\[\ce{C2H5Br ->[Na/Dry ether] C2H5 - C2H5}\]
 A product (1) of above reaction is:-
A product (1) of above reaction is:-
Grignard reagent shows addition on
The product formed in the first step of the reaction
\[\begin{array}{cc}
\ce{Br}\phantom{......}\\
|\phantom{.......}\\
\ce{CH3-CH2-CH-CH2-CH-CH3}\\
\phantom{...............}|\\
\phantom{................}\ce{Br}
\end{array}\]
with excess Mg/Et2O(Et = C2H5) is:
Explain why Grignard reagents should be prepared under anhydrous conditions.
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
