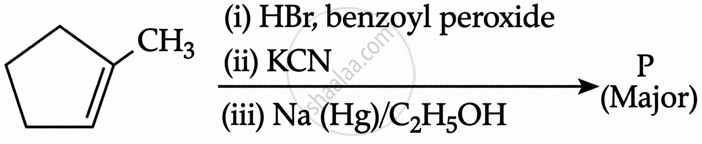Advertisements
Advertisements
प्रश्न
How will you bring about the following conversion?
Ethanol to ethyl fluoride
How will you bring about the following conversion in not more than two steps?
Ethanol to Ethyl fluoride
Advertisements
उत्तर
\[\ce{\underset{Ethanol}{CH3CH2OH} ->[SOCl2, Pyridine][-SO2, -HCl] \underset{Ethyl chloride}{CH3CH2Cl} ->[Hg2F2][-Hg2Cl2] \underset{Ethyl fluoride}{CH3CH2F}}\]
APPEARS IN
संबंधित प्रश्न
Why is sulphuric acid not used during the reaction of alcohols with KI?
A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
Write the equation for the preparation of 1-iodobutane from 1-butanol.
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2Cl + NaI ->[acetone][heat]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH = CH2 + HBr->[peroxide]}\]
Primary alkyl halide C4H9Br (a) reacted with alcoholic KOH to give compound (b). Compound (b) is reacted with HBr to give (c) which is an isomer of (a). When (a) is reacted with sodium metal it gives compound (d), C8H18 which is different from the compound formed when n-butyl bromide is reacted with sodium. Give the structural formula of (a) and write the equations for all the reactions.
How the following conversion can be carried out?
Benzyl alcohol to 2-phenylethanoic acid
How the following conversion can be carried out?
But-1-ene to n-butyliodide
How the following conversion can be carried out?
tert-Butyl bromide to isobutyl bromide
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields three isomeric monochlorides.
Which is the incorrect option?
3-Methyl-pent-2-ene of peroxide forms an addition product. The number of possible stereoisomers for the product is ______.
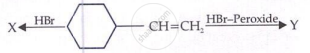
X and Y will be?
The reagent used in the conversion of 1-butanol to 1-bromobutane is ______.
Predict the major product ‘P’ in the following sequence of reactions: