Advertisements
Advertisements
Question
Write a short note on Fittig reaction.
Chemical Equations/Structures
Short/Brief Note
Advertisements
Solution
- The Fittig reaction is a coupling reaction between two aryl halides.
- It involves the use of sodium metal to facilitate the reaction.
- Under reaction conditions, sodium removes the halogen atoms from the aryl halides.
- This results in the formation of a covalent bond between two aromatic rings.
- The reaction produces symmetrical biaryl compounds (two identical aromatic rings joined).
- It typically requires heating and anhydrous conditions.
- The mechanism proceeds through the formation of aryl radicals or aryl sodium intermediates.
- The Fittig reaction is important for synthesizing biphenyls and related aromatic compounds.
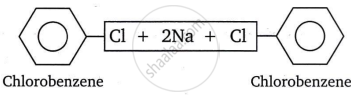
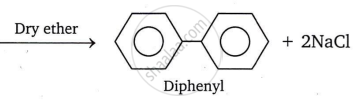
- When a haloarene is heated with sodium metal in the presence of dry ether, the halogen atom is replaced by an aryl group, yielding a diaryl. This reaction is known as the Fittig reaction.
For example:
shaalaa.com
Is there an error in this question or solution?
