Advertisements
Advertisements
Question
Which compound in the following pair will react faster in SN2 reaction with OH−?
(CH3)3CCl or CH3Cl
Advertisements
Solution
\[\begin{array}{cc}
\phantom{......}\ce{CH3}\\
\phantom{...}|\\
\ce{CH3 - C - Cl}\\
\phantom{...}|\\
\phantom{......}\ce{CH3}\\
\end{array}\] or CH3, Cl
The SN2 mechanism involves attack by the nucleophile at the carbon bearing the leaving group. But, in the case of (CH3)3CCl, the attack of the nucleophile at the carbon atom is hindered because of the presence of bulky substituents on that carbon atom bearing the leaving group. On the other hand, there are no bulky substituents on the carbon atom bearing the leaving group in CH3Cl. Hence, CH3Cl reacts faster than (CH3)3CCl in SN2 reaction with OH−.
APPEARS IN
RELATED QUESTIONS
Write the major products(s) in the following:

Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Which would undergo SN2 reaction faster in the following pair and why ?

In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

What happens when methyl chloride is treated with KCN?
SN1 reactions are accompanied by racemization in optically active alkyl halides.
What is the action of the following on ethyl bromide:
moist silver oxide
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Which of the following is optically inactive?
Which of the following is the correct order of decreasing SN2 reactivity?
An organic molecule necessarily shows optical activity if it ____________.
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
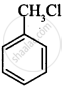
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
Chlorination of alkanes is an example of
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
Retention of configuration is observed in ______.
