Advertisements
Advertisements
Question
Which compound in the following pair will react faster in SN2 reaction with OH−?
CH3Br or CH3I
Advertisements
Solution
In the SN2 mechanism, the reactivity of halides for the same alkyl group increases in the order. This happens because, as the size increases, the halide ion becomes a better leaving group.
R−F << R−Cl < R−Br < R−I
Therefore, CH3I will react faster than CH3Br in SN2 reactions with OH−.
APPEARS IN
RELATED QUESTIONS
Write the major products(s) in the following:

Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
CH3CH2CH2CH2Br or \[\begin{array}{cc}
\ce{CH3CH2CHCH3}\\
\phantom{.....}|\\
\phantom{.......}\ce{Br}\
\end{array}\]
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
What is the action of the following on ethyl bromide:
moist silver oxide
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Which one is most reactive towards SN1 reaction?
Most reactive halide towards SN1 reaction is ____________.
In the SN1 reaction, racemization takes place. It is due to:
Which of the following is the correct order of decreasing SN2 reactivity?
Among the following, the dissociation constant is highest for:
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
Complete the following analogy:
Same molecular formula but different structures: A : : Non superimposable mirror images: B
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Chlorination of alkanes is an example of
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
In which reaction mechanism carbocation is formed?
Discuss the mechanism of alkaline hydrolysis of methyl bromide.
Explain why Grignard reagents should be prepared under anhydrous conditions.
The correct order of increasing reactivity of
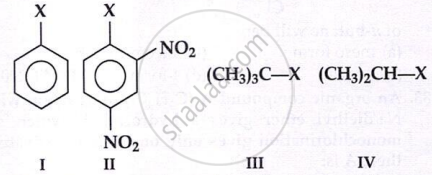
C-X bond towards nucleophile in the following compounds is:
