Advertisements
Advertisements
Question

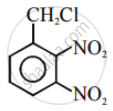
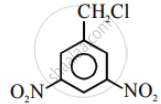
Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Advertisements
Solution
(i) (a) and (e) both are nucleophiles.
(iii) In (c) carbon atom is sp2 hybridised.
Explanation:
HO− and Cl− are nucleophiles.
In (iii), C atom is sp2 hybridised due to formation of \[\ce{C - OH}\] bond and breaking of \[\ce{C - Cl}\] bond simultaneously. So, in the transition state, the C atom is bonded to only 3 H atoms completely.
APPEARS IN
RELATED QUESTIONS
Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction
How do you convert the following:
Ethanol to propanenitrile
Write the structure of the major product in each of the following reaction :

The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
What happens when chlorobenzene is subjected to hydrolysis?
What happens when methyl chloride is treated with KCN?
AgCN reacts with haloalkanes to form isocyanide. Haloalkanes react with KCN to form alkyl cyanides as the main product. Why?
Which among MeX, RCH2X, R2CHX and R3CX is most reactive towards SN2 reaction?
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
CCl4 is insoluble in water because:-
In which reaction mechanism carbocation is formed?
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.
Inversion of configuration occurs in ______.
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
Which one of the following chlorohydrocarbons readily undergoes solvolysis?