Advertisements
Advertisements
Question
Which is the correct increasing order of boiling points of the following compounds?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Options
Bromobenzene < 1-Bromobutane < 1-Bromopropane < 1-Bromoethane
Bromobenzene < 1-Bromoethane < 1-Bromopropane < 1-Bromobutane
1-Bromopropane < 1-Bromobutane < 1-Bromoethane < Bromobenzene
1-Bromoethane < 1-Bromopropane < 1-Bromobutane < Bromobenzene
Advertisements
Solution
1-Bromoethane < 1-Bromopropane < 1-Bromobutane < Bromobenzene
Explanation:
The attractions get stronger as the molecules get bigger in size and have more electrons.
APPEARS IN
RELATED QUESTIONS
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
Explain why alkyl halides, though polar, are immiscible with water?
Why dextro and laevorotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
The decreasing order of boiling points of alkyl halides is:
Which of the following is liquid at room temperature (b.p. is shown against it)?
Which of the following possesses the highest melting point?
Arrange the following compounds in the increasing order of their densities.
(a)

(b)

(c)
(d)

Arrange the following compounds in increasing order of their boiling points.
(a) \[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
\backslash\phantom{...........}\\
\ce{CH - CH - CH2Br}\\
/\phantom{.............}\\
\ce{CH3}\phantom{..................}
\end{array}\]
(b) \[\ce{CH3CH2CH2CH2Br}\]
(c) \[\begin{array}{cc}
\phantom{...}\ce{CH3}\\
\phantom{}|\\
\ce{H3C - C - CH3}\\
\phantom{}|\\
\phantom{..}\ce{Br}
\end{array}\]
Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
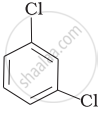
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound:
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
