Advertisements
Advertisements
Question
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
Advertisements
Solution
The boiling point of n-butyl bromide is higher than that of t-butyl bromide because n-butyl bromide is a straight chain molecule having larger surface area and therefore, has stronger intermolecular forces. On the other hand, t-butyl bromide is branched molecule, so it has a smaller surface area. Hence, it has weaker intermolecular force.
APPEARS IN
RELATED QUESTIONS
Which of the following possesses the highest melting point?
Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
 |
| (III) | 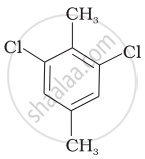 |
Assertion: The boiling points of alkyl halides decrease in the order:
RI > RBr > RCl > RF
Reason: The boiling points of alkyl chlorides, bromides and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
Which out of the following is an intensive property?
Why alkyl halides though polar are immiscible with water?
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound:
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
