Advertisements
Advertisements
Question
Arrange the following compounds in the increasing order of their densities.
(a)

(b)

(c)
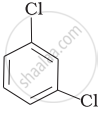
(d)

Options
(a) < (b) < (c) < (d)
(a) < (c) < (d) < (b)
(d) < (c) < (b) < (a)
(b) < (d) < (c) < (a)
Advertisements
Solution
(a) < (b) < (c) < (d)
Explanation:
The density increases with increase in number of carbon atoms, halogen atoms and atomic masses of the halogen atoms bounded with the alkyl or aryl groups. Or, in other words density is related to molecular mass of the compound. Higher the molecular mass, greater will be the density of the compound.
APPEARS IN
RELATED QUESTIONS
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
Define racemic mixture.
Why dextro and laevorotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Which of the following compounds has the highest boiling point?
Which of the following halide is 2°?
The decreasing order of boiling points of alkyl halides is:
Which of the following is liquid at room temperature (b.p. is shown against it)?
Mg reacts with RBr best in ____________.
Which is the correct increasing order of boiling points of the following compounds?
1-Iodobutane, 1-Bromobutane, 1-Chlorobutane, Butane
Which is the correct increasing order of boiling points of the following compounds?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Out of o-and p-dibromobenzene which one has higher melting point and why?
Why alkyl halides though polar are immiscible with water?
Why is the boiling point of o-dichlorobenzene higher than p-dichlorobenzene, but the melting point of para-isomer is higher than ortho-isomer?
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound:
4-tert-Butyl-3-iodoheptane
