Advertisements
Advertisements
Question
Out of o-and p-dibromobenzene which one has higher melting point and why?
Advertisements
Solution
p-Dibromobenzenc has higher melting point than its o-isomer.
It is due to symmetry of p-isomer which fits in crystal lattice better than the o-isomer.
APPEARS IN
RELATED QUESTIONS
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
Arrange the set of compounds in order of increasing boiling points.
1-Chloropropane, Isopropyl chloride, 1-Chlorobutane.
Explain why alkyl halides, though polar, are immiscible with water?
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
Which of the following halide is 2°?
The decreasing order of boiling points of alkyl halides is:
p-dichlorobenzene has a higher melting point than its o- and m-isomers because ____________.
Arrange the following compounds in the increasing order of their densities.
(a)

(b)

(c)
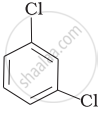
(d)

Arrange the following compounds in increasing order of their boiling points.
(a) \[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
\backslash\phantom{...........}\\
\ce{CH - CH - CH2Br}\\
/\phantom{.............}\\
\ce{CH3}\phantom{..................}
\end{array}\]
(b) \[\ce{CH3CH2CH2CH2Br}\]
(c) \[\begin{array}{cc}
\phantom{...}\ce{CH3}\\
\phantom{}|\\
\ce{H3C - C - CH3}\\
\phantom{}|\\
\phantom{..}\ce{Br}
\end{array}\]
Assertion: The boiling points of alkyl halides decrease in the order:
RI > RBr > RCl > RF
Reason: The boiling points of alkyl chlorides, bromides and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
Which out of the following is an intensive property?
Why alkyl halides though polar are immiscible with water?
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3 C(C2H5)2CH2Br}\]
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
