Advertisements
Advertisements
Question
Identify 'A' in the following reaction -

(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
Advertisements
Solution
1 -Bromo-2,2-dimethylpropane

APPEARS IN
RELATED QUESTIONS
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Write the structure of the major product in each of the following reaction :

What happens when methyl chloride is treated with KCN?
How the following conversion can be carried out?
Ethyl chloride to propanoic acid.
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
In the SN1 reaction, racemization takes place. It is due to:
SN2 mechanism proceeds through intervention of ____________.
Which among MeX, RCH2X, R2CHX and R3CX is most reactive towards SN2 reaction?
Isopropyl chloride undergoes hydrolysis by:
An organic molecule necessarily shows optical activity if it ____________.
Which of the following statements are correct about the kinetics of this reaction?

(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
Compound ‘A’ with molecular formula \[\ce{C4H9Br}\] is treated with aq. \[\ce{KOH}\] solution. The rate of this reaction depends upon the concentration of the compound ‘A’ only. When another optically active isomer ‘B’ of this compound was treated with aq. \[\ce{KOH}\] solution, the rate of reaction was found to be dependent on concentration of compound and \[\ce{KOH}\] both.
(i) Write down the structural formula of both compounds ‘A’ and ‘B’.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
How do polar solvents help in the first step in SN1 mechanism?
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Which of the following is the definition of chirality?
Convert bromoethane to propanamine.
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
Identify the product in the following reaction: 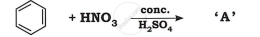
Discuss SN2 mechanism of methyl bromide using aqueous KOH.
Which of the following is halogen exchange reaction?
The correct order of increasing reactivity of
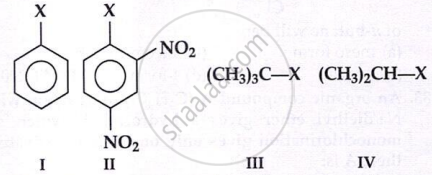
C-X bond towards nucleophile in the following compounds is:
The compound that will undergo SN1 reaction with the fastest rate is:
