Advertisements
Advertisements
Question
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
Advertisements
Solution
\[\begin{array}{cc}\ce{C2H5 - CH - CH3}\\
|\phantom{.}\\\ce{Br}\end{array}\]
Mechanism:
\[\begin{array}{cc}
\ce{H}\phantom{.................}\ce{H}\\
|\phantom{..................}|\\
\ce{H3C - C - C2H5 ->[Slow step] C^⊕ + B\overset{⊖}{r}}\\
\phantom{...}|\phantom{................}/\phantom{...}\backslash\\
\phantom{.......}\ce{Br}\phantom{...........}\ce{H3C}\phantom{....}\ce{C2H5}
\end{array}\]
\[\begin{array}{cc}
\ce{H}\phantom{....................}\ce{H}\phantom{...............}\ce{H}\phantom{.......}\\
|\phantom{.....................}|\phantom{................}|\phantom{.......}\\
\ce{C^⊕ + O\overset{⊖}{H} ->[Fast] H3C - C - OH + HO - C - CH3}\\
/\phantom{..}\backslash\phantom{....................}|\phantom{.................}|\phantom{.........}\\
\ce{H3C}\phantom{...}\ce{C2H5}\phantom{...............}\ce{C2H5}\phantom{.............}\ce{C2H5}\phantom{........}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Which would undergo SN1 reaction faster in the following pair and why?

Which one is most reactive towards SN1 reaction?
Which of the following is the correct order of decreasing SN2 reactivity?
Which of the following is an optically active compound?
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
Complete the reaction with the main product formed:

The correct order of increasing reactivity of
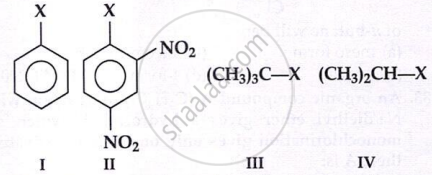
C-X bond towards nucleophile in the following compounds is:
Assertion (A):  undergoes SN2 reactions faster than
undergoes SN2 reactions faster than  .
.
Reason (R): Iodine is a better leaving group because of its large size.
In the light of the above statements, choose the correct answer from the options given below:
