Advertisements
Advertisements
Question
Assertion (A) : Nucleophilic substitution of iodoethane is easier than chloroethane.
Reason (R) : Bond enthalpy of C-I bond is less than that of C-Cl bond.
Options
Both (A) and (R) are true and (R) is the correct explanation of (A).
Both (A) and (R) are true, but (R) is not the correct explanation of (A).
(A) is true, but (R) is false.
(A) is false, but (R) is true.
Advertisements
Solution
Both (A) and (R) are true and (R) is the correct explanation of (A).
Explanation:
Iodine is a better leaving group than chlorine because of its larger size, it will be released at a higher rate in the presence of an incoming nucleophile.
APPEARS IN
RELATED QUESTIONS
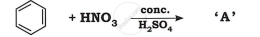
Write the structures of A, B and C in the following:

Which compound in the following pair will react faster in SN2 reaction with OH−?
(CH3)3CCl or CH3Cl
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
Which of the following pairs is/are correctly matched?
| Reaction | Product | |
| I | RX + AgCN | RNC |
| II | RX + KCN | RCN |
| III | RX + KNO2 | \[\begin{array}{cc} \phantom{.......}\ce{O}\\ \phantom{.....}/\\ \ce{R - N}\phantom{....}\\ \phantom{.....}\backslash\backslash\\ \phantom{.......}\ce{O} \end{array}\] |
| IV | RX + AgNO2 | \[\ce{R-O-N=O}\] |
Most reactive halide towards SN1 reaction is ____________.
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Give reason for the following:
The product formed during SN1 reaction is a racemic mixture.
The number of chiral carbons present in the molecule given below is ______.

Identify the product in the following reaction: