Advertisements
Advertisements
Question
What happens when chlorobenzene is subjected to hydrolysis?
Advertisements
Solution
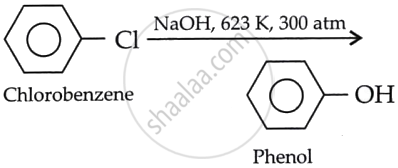
Chlorobenzene does not undergo hydrolysis under normal conditions. However, it undergoes hydrolysis when heated in an aqueous sodium hydroxide solution at 623 K and 300 atm, forming phenol. The presence of an electron-withdrawing group increases the reactivity of haloarenes.
APPEARS IN
RELATED QUESTIONS
Write the major products(s) in the following:

Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq{.} ethanol]}\]
What is the action of the following on ethyl bromide?
silver acetate
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Which compound in the following pair reacts faster in SN2 reaction with OH–?
- CH3Br or CH3
- CH3Cl, (CH3)3CCl
Which of the following is the correct order of decreasing SN2 reactivity?
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Among the following, the dissociation constant is highest for:
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.

(I)

(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
The reaction of C6H5–CH=CH–CH3 with HBr produces:
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Which one of the following compounds is more reactive towards SN1 reaction?
The number of chiral carbons present in the molecule given below is ______.

Optical activity of an enantiomeric mixture is +12.6° and the specific rotation of (+) isomer is +30°. The optical purity is ______ %.
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}
\ce{CH3CHCH2CH2Br}\\
|\phantom{.............}\\
\ce{CH3}\phantom{..........}\\
\end{array}\] or \[\begin{array}{cc}
\ce{CH3CH2CHCH2Br}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]
The compound that will undergo SN1 reaction with the fastest rate is:
